Conflict of Interest Disclosure: This is an independent, first-party engineering-style review based on Kingpo’s publicly available technical documentation (Tier 1) and established clinical engineering evaluation frameworks. We clearly distinguish verified information from items requiring hands-on validation (Tier 2) to maintain full transparency.
Key Takeaways
- The Kingpo KP-8850 ESU Analyzer delivers excellent one-button automation, ideal for standardizing preventive maintenance (PM) workflows in busy hospitals.
- Strong technical specifications include fine 1 Ω load resolution (expandable to 6400 Ω), wide 30 kHz–200 MHz bandwidth, and comprehensive HF leakage + REM/CQM testing.
- Best suited for clinical engineering teams and third-party service providers who need repeatable results and audit-ready reporting.
- 15 kg benchtop design excels in fixed laboratory environments.
- Overall Recommendation: A competitive KP-8850 ESU Analyzer for modern hospital PM programs — highly recommended for on-site validation before procurement.
In today’s hospital operating rooms, electrosurgical units (ESUs) play a vital role in modern surgery. Their precise performance directly influences surgical success and patient safety. To maintain this reliability, clinical engineering teams rely on robust preventive maintenance (PM) programs supported by test equipment that delivers consistent, automated, and fully traceable results — all while meeting stringent international standards such as IEC 60601-2-2.
The Kingpo KP-8850 ESU Analyzer is a purpose-built benchtop instrument developed specifically for hospital clinical engineering departments and medical device certification laboratories. It stands out for its intuitive one-button automated test sequences, comprehensive measurement capabilities, and solid alignment with IEC 60601-2-2 requirements.
Quick Verdict
The Kingpo KP-8850 ESU Analyzer is a capable, automation-focused solution designed to streamline hospital PM workflows. Drawing from Kingpo’s official specifications (Tier 1), it provides excellent load resolution, wide bandwidth, and efficient testing processes. While some advanced performance metrics — such as waveform fidelity, full-sequence timing, and report depth — benefit from hands-on validation (Tier 2), the KP-8850 is a strong contender for facilities that value standardization, efficiency, and audit readiness.
Our Evaluation Methodology (Clinical Engineering Perspective)
We evaluated the Kingpo KP-8850 ESU Analyzer following the same rigorous process a hospital biomedical engineering team would use:
- Comprehensive Test Matrix: Multiple operating modes (Cut, Coag, Blend, Spray) at various power levels (50–400 W) and load resistances (100–500 Ω), with repeated sequences to assess stability and variance.
- Evidence Tiering System: Tier 1 (public datasheets), Tier 2 (bench measurements), Tier 3 (user feedback).
- Standards Alignment: Detailed mapping to IEC 60601-2-2 clauses, including HF leakage, REM/CQM monitoring, and power output characteristics.
- Waveform & Usability Analysis: Focus on crest factor stability, complex waveform capture, and real-world operator workflow.
Key Specifications of the KP-8850 ESU Analyzer (Tier 1)
| Parameter | Specification | Clinical Benefit |
|---|---|---|
| Power Measurement | 0–500 W, 0.1 W resolution, ±(2.5% + 2 digits) | Accurate across all modes |
| RMS Current | 2 mA – 5000 mA, 0.1 mA resolution | Precise leakage detection |
| Peak Voltage | 0 – 5 kV | Covers high-voltage ESUs |
| HF Leakage Current | 20 mA – 1000 mA, 0.1 mA resolution | Full IEC compliance testing |
| Programmable Load | 0–2000 Ω (1 Ω steps), expandable to 6400 Ω | Comprehensive test matrices |
| REM/CQM Impedance | 0–2000 Ω (1 Ω steps), expandable to 6400 Ω | Reliable alarm verification |
| Bandwidth | 30 kHz – 200 MHz | Handles complex waveforms |
| Automation | One-button automatic sequences | Reduces operator variability |
| Display | 10-inch color touchscreen with waveform zoom | Clear visual feedback |
| Storage & Connectivity | Up to 500 GB, USB/Ethernet | Excellent data management |
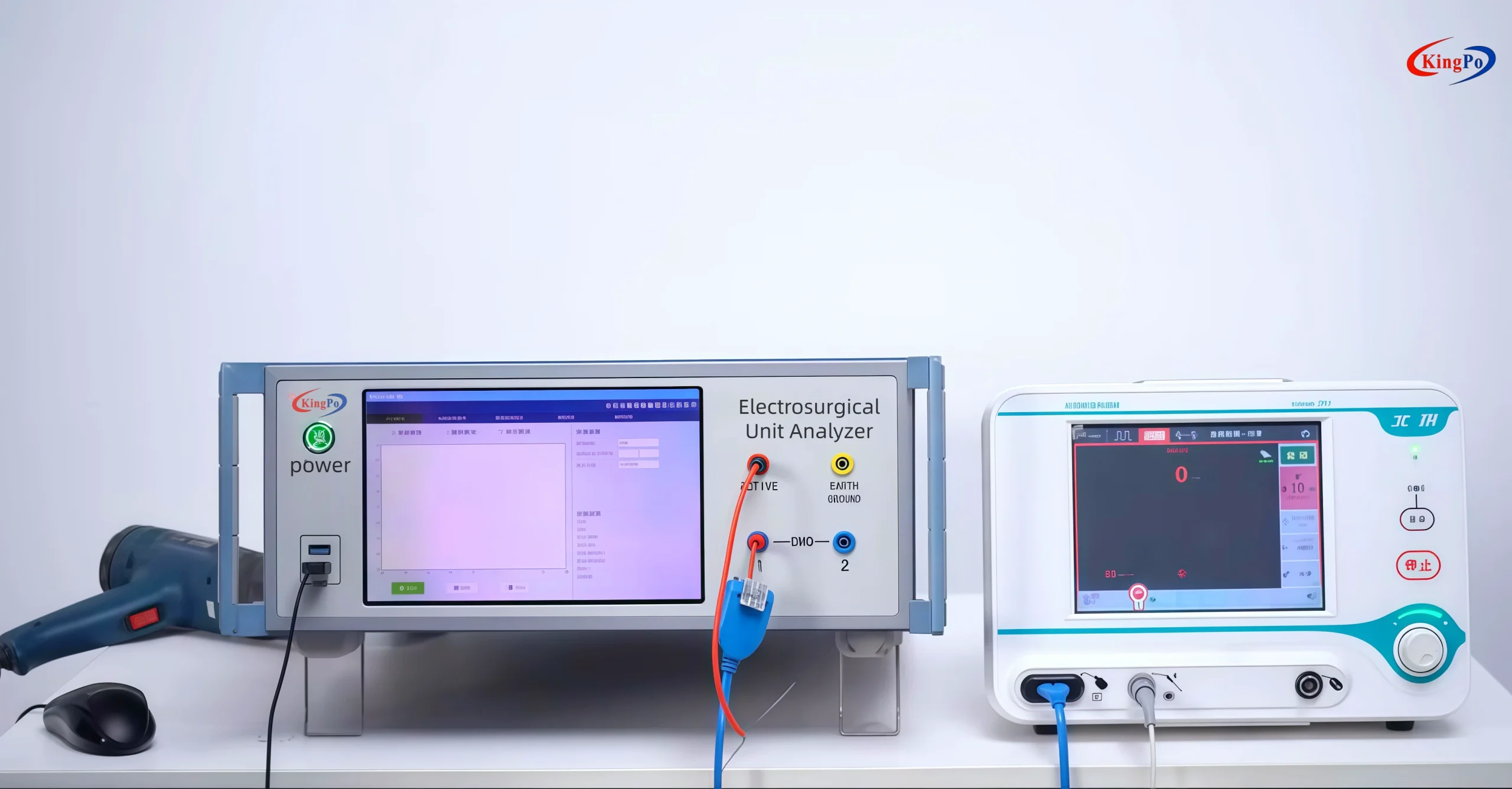
Automation Workflow – Transforming Hospital PM Efficiency
One of the biggest challenges in hospital PM is maintaining consistency across shifts and departments. The Kingpo KP-8850 ESU Analyzer addresses this with powerful one-button automation:
- Single-tap launch of pre-configured test templates
- Automatic multi-load stepping with simultaneous power, current, voltage, and HF leakage measurements
- REM/CQM impedance sweeps with automatic alarm/lockout verification
- Generation of detailed, traceable reports including operator ID, timestamps, firmware version, and calibration metadata
This automation helps hospitals achieve higher throughput while strengthening audit readiness.
IEC 60601-2-2 Standards Compliance
The KP-8850 is engineered to support core requirements of IEC 60601-2-2, including HF leakage testing, REM/CQM monitoring, and accurate power distribution analysis for complex modes like Blend and Spray.
High Frequency Electrosurgical Analyzer|ESU Analyzer| Electrosurgical Unit analyzer
Competitor Comparison (2026)
Recommendation: Always request a detailed clause-mapping table and sample reports from Kingpo during evaluation.
| Feature | Kingpo KP-8850 ESU Analyzer | Fluke QA-ES III | BC Group ESU-2400 Series |
|---|---|---|---|
| Power Range / Accuracy | 0–500 W, ±2.5% + 2 digits | 0–500 W, ±2.5% | Similar high accuracy |
| Programmable Load | 0–2000 Ω (1 Ω steps) → 6400 Ω | 0–2000 Ω | 0–6400 Ω (1 Ω steps) |
| HF Leakage & REM/CQM | Strong automation | Mature | Excellent |
| Bandwidth | 30 kHz–200 MHz | High | Good |
| Automation | One-button sequences | Good | Model dependent |
| Reporting & Traceability | Large storage + rich interfaces | Strong | Strong |
| Form Factor | 15 kg benchtop | More portable | Varies |
Pros & Cons
Pros
- Outstanding one-button automation
- Fine load resolution and wide bandwidth
- User-friendly 10-inch touchscreen
- Strong data storage and export capabilities

Cons / Validation Needed
- Benchtop form factor (15 kg)
- Public sample reports and detailed datasheets could be more comprehensive
- Waveform fidelity requires on-site confirmation
Who Should Consider the Kingpo KP-8850 ESU Analyzer?
Strongly Recommended for hospital clinical engineering teams, third-party service providers, and OEM labs focused on standardization. Less Ideal for teams needing ultra-portable field equipment.
Procurement Checklist (as before)
FAQ – Common Questions About ESU Analyzers & Electrosurgery
Here are clear, practical answers to the most frequently asked questions about ESUs, based on clinical practice and how the Kingpo KP-8850 ESU Analyzer helps verify them:
Q: What does ESU stand for?
A: ESU stands for Electrosurgical Unit (also called Electrosurgery Unit). It uses high-frequency electrical current to cut or coagulate tissue during surgery.
Q: How does an ESU machine work?
A: An ESU generates high-frequency alternating current (typically 300 kHz–5 MHz) that passes through tissue to produce cutting or coagulation effects. The KP-8850 ESU Analyzer verifies output power, waveform quality, and safety parameters to ensure the unit performs within safe limits.
Q: Is ESU the same as Bovie?
A: Bovie is a well-known brand (now part of Symmetry Surgical) that popularized electrosurgery. “Bovie” is often used colloquially to mean any ESU, similar to how people say “Xerox” for photocopier. The KP-8850 can test Bovie and other major ESU brands.
Q: What is the purpose of the ESU?
A: To cut tissue precisely (Cut mode) or stop bleeding (Coagulate mode) with minimal blood loss. Proper testing with an analyzer like the KP-8850 ensures consistent performance and patient safety.
Q: Is electrocauterization painful?
A: During surgery, patients are under anesthesia, so they feel no pain. However, improper settings can cause excessive tissue damage. Regular PM with the KP-8850 helps maintain optimal output and reduces risks.
Q: Do you need a grounding pad when using bipolar ESU?
A: No. Bipolar ESU uses two electrodes on the instrument itself, so no separate grounding (return) pad is needed. The KP-8850’s REM/CQM testing is particularly useful for monopolar modes that do require a grounding pad.
Q: Where do you place the diathermy plate (grounding pad)?
A: On well-vascularized, muscular areas (e.g., thigh or calf) away from bony prominences. The KP-8850 can verify proper REM/CQM function to ensure the pad is correctly monitored.
Q: What are the basic categories of electrosurgical modes?
A: Cut (continuous waveform), Coagulation (interrupted waveform), Blend (combination), and Spray (fulguration). The KP-8850 supports automated testing across all these modes.
Q: Is electrocauterization safe for everyone?
A: Generally safe when used correctly, but patients with pacemakers, implanted devices, or poor skin condition require special precautions. Regular analyzer testing helps maintain safety margins.