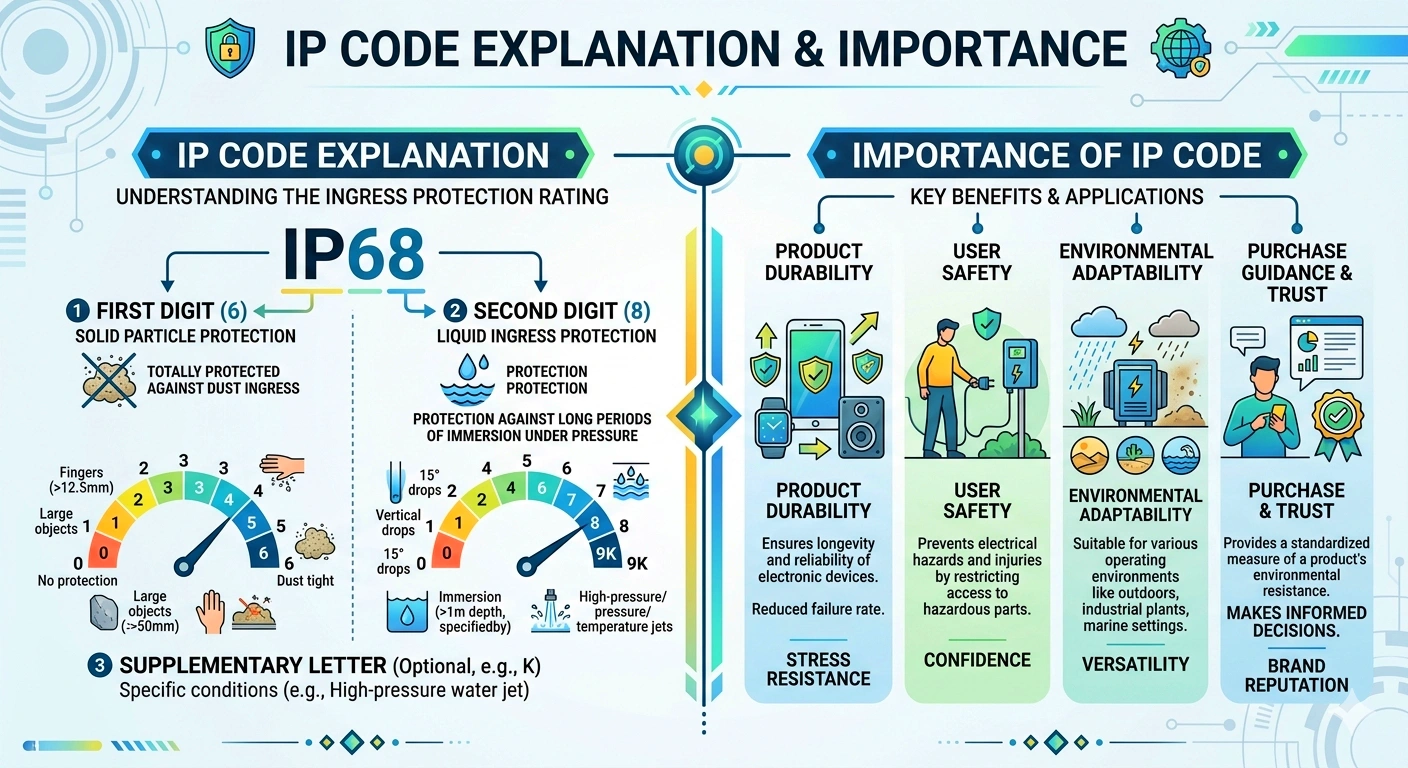
ISO 80369-7 Figure C.2 Male Luer Lock Reference Connector
- Meets ISO 80369 dimensional requirements.
- Replaces legacy ISO 594 and ISO 594-1 standards.
- Improves patient safety via non-interconnectability.
- Ideal for intravascular or hypodermic applications.
Advanced Gauges for Small Bore Connectors
Following the ISO 80369-1 general requirements, our series provides specialized testing solutions for liquids and gases in healthcare. These small bore connectors intended for clinical use are verified using high-precision male luer lock reference gauges to prevent cross-connection risks.
Industrial & Clinical Applications
Dimensional Verification
Validating the dimensional requirements of female luers during medical device manufacturing.
Risk Mitigation
Ensuring patient safety by verifying non-interconnectable designs in gases in healthcare applications.
Regulatory Compliance
Helping manufacturers meet 80369-7 compliant standards for global regulatory audits.
Related ISO 80369 Standards Matrix
| Standard | Clinical Application | Focus Area |
|---|---|---|
| ISO 80369-3 | Enteral Applications | Feeding Tubes & Syringes |
| ISO 80369-6 | Neuraxial Applications | Regional Anesthesia |
| ISO 80369-7 | Intravascular / Hypodermic | Luer Connector Systems |
Frequently Asked Questions
How does ISO 80369-7 improve patient safety?
By mandating unique dimensional requirements, it ensures that small bore connectors from different clinical categories (like enteral vs intravascular) cannot be cross-connected.
Is Figure C.2 compatible with ISO 594-1?
While ISO 80369-7 supersedes ISO 594-1, our connectors are built to the latest 80369-7 compliant specs to ensure future-proof testing for all medical devices.
Ensure Your Dimensional Compliance Today
Get a technical quote for NIST-traceable ISO 80369-7 Figure C.2 reference connectors.