





ISO 18193 Figure C.2 Vascular Model | Dual Lumen Cannula Recirculation Testing
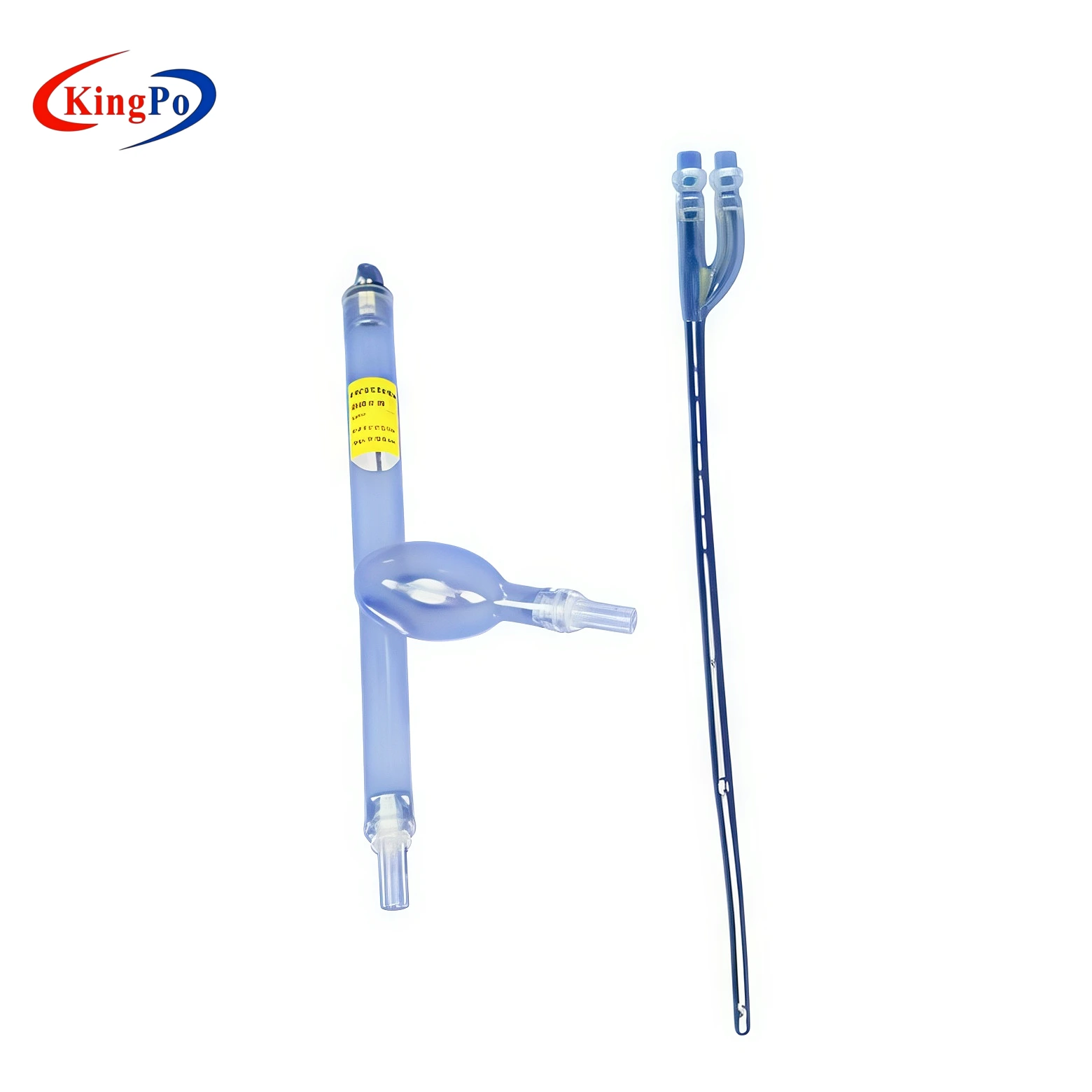
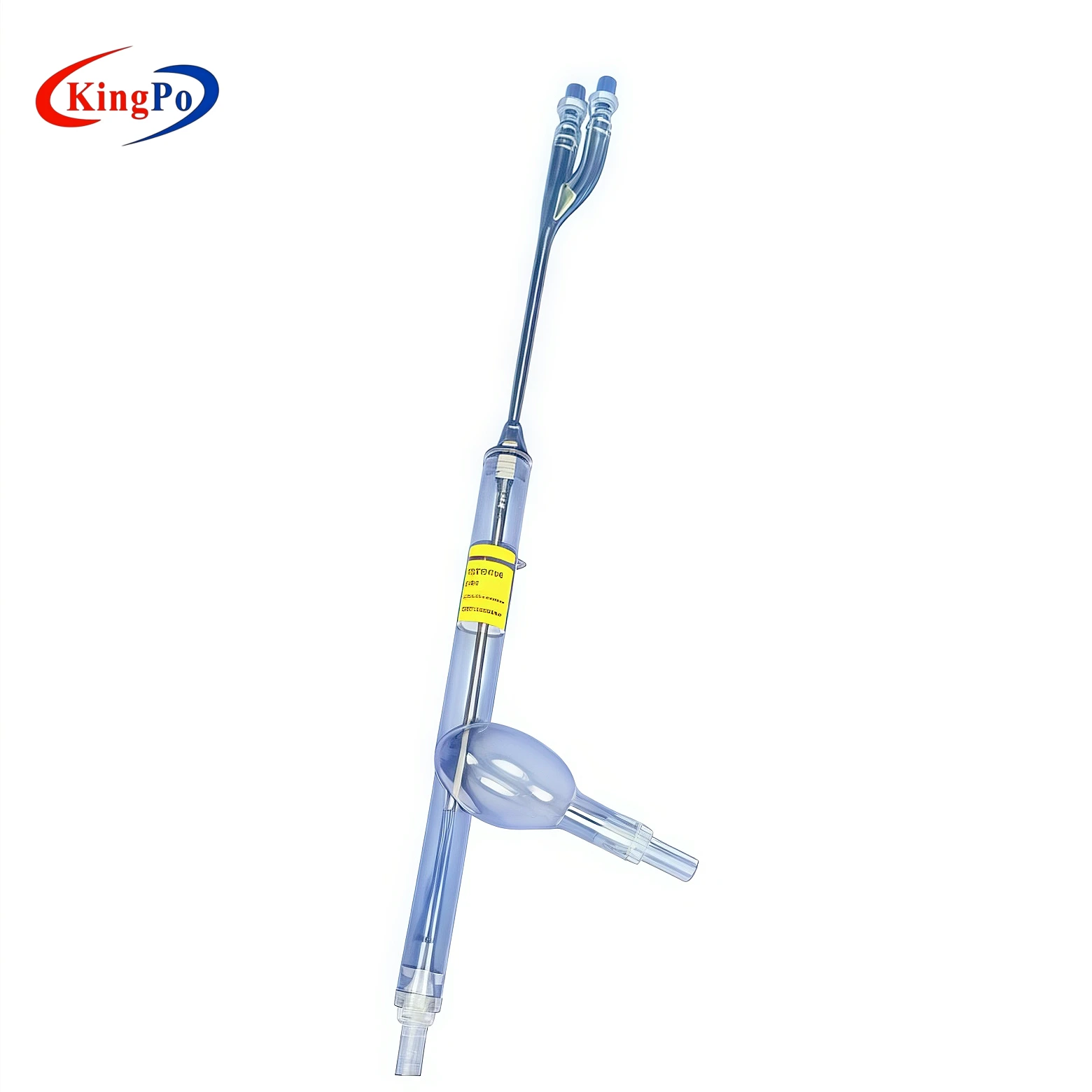
The ISO 18193 Figure C.2 Vascular Model is a high-fidelity anatomical test fixture that simulates the superior vena cava, right atrium and inferior vena cava for recirculation performance testing of dual-lumen cannulae in extracorporeal circulation systems.
- Standard Compliance: ISO 18193 Figure C.2 – exact vascular model for dual-lumen cannula recirculation testing
- Anatomical Regions: Superior vena cava, right atrium, inferior vena cava
- Test Medium: Ink or particles for visual recirculation assessment
- Application: Recirculation testing of dual-lumen cannulae in CPB, ECLS, ECCO₂R
- Material: High-clarity medical-grade transparent model
- Compatibility: Standard extracorporeal test circuits
- MOQ: 1
- Delivery Period: 7 working days
ISO 18193 Figure C.2 Vascular Model Description
ISO 18193 Figure C.2 Vascular Model | Dual Lumen Cannula Recirculation Testing
The ISO 18193 Figure C.2 Vascular Model | Dual Lumen Cannula Recirculation Testing is a professional anatomical simulation device manufactured by KingPo. It is specifically designed to replicate the superior vena cava, right atrium and inferior vena cava for recirculation testing of dual-lumen cannulae used in extracorporeal circulation, in full accordance with ISO 18193 Figure C.2 and Annex C.
Technical Parameters
| Parameter | Specification | Remark / Notes |
|---|---|---|
| Standard | ISO 18193 Figure C.2 | Vascular model for dual-lumen cannula recirculation testing |
| Anatomical Regions | Superior vena cava, right atrium, inferior vena cava | Exact geometry per ISO 18193 Annex C |
| Test Medium | Ink or particles | Visualizes recirculation paths in circuit |
| Application | Dual-lumen cannula testing | Extracorporeal circulation (CPB, ECLS, ECCO₂R, etc.) |
| Material | High-clarity medical-grade transparent model | Allows direct observation of flow and recirculation |
| Certification | ISO 17025 traceable | Supports laboratory accreditation and audit requirements |
The vascular model is precision-molded from high-clarity medical-grade material to exactly replicate the internal geometry of the superior vena cava, right atrium and inferior vena cava as defined in ISO 18193 Figure C.2. All critical vessel diameters, curvatures and junction angles are manufactured to tight tolerances to ensure repeatable flow dynamics and accurate recirculation visualization.
The transparent construction allows direct observation of ink or particle paths during testing, while the rigid yet anatomically compliant structure maintains dimensional stability under typical test pressures and flow rates. Mounting points are integrated for secure attachment to standard test circuits without flow distortion.
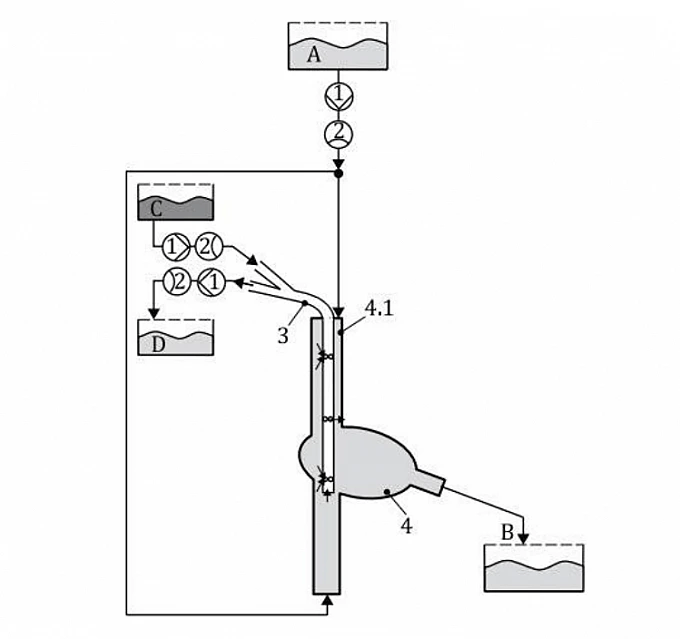
Key: 1 rotary blood pump 2 flow sensor 3 cannula under test 4 vamvcaiafi A reservoir A (inlet reservoir of body circulation) B reservoir B (outlet reservoir of body circulation) c reservoir C (inlet reservoir of test circulation through dual-lumen canula) D reservoir D (outlet reservoir of test circulation through dual-lumen canula)
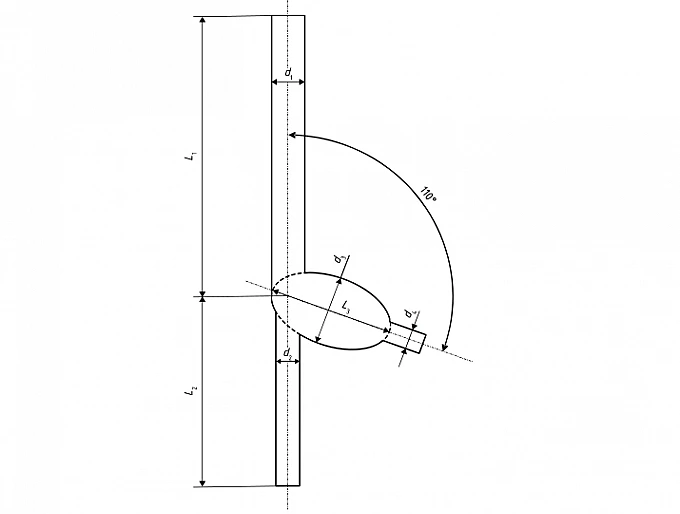
Key: Ddiameter of device under test di=2,65x D d2=2xD d3=5,63×D d4 =1,6xD L1 =22,5× D L2=15,6×D L3 =10xD
Testing Principle
The model is integrated into a closed extracorporeal circuit with a dual-lumen cannula. Controlled blood-analogue flow is introduced through the cannula while ink or particles are injected to visualize and quantify recirculation between the drainage and return lumens. The test evaluates the degree of recirculation under simulated physiological conditions as specified in ISO 18193 Annex C.
Common Error Mitigation
To ensure repeatable and accurate results, always verify proper cannula insertion depth and alignment within the model before starting the test. Use a consistent blood analogue viscosity and maintain stable flow rates. Regular flushing and visual inspection of the model interior prevent particle residue that could affect flow visualization or lead to false recirculation readings.
Primary Use Cases & Business Value
The ISO 18193 Figure C.2 Vascular Model is used by medical device manufacturers and certification laboratories to evaluate recirculation performance of dual-lumen cannulae. It provides objective, standardized data essential for product development, quality control and regulatory approval of cannulae used in cardiopulmonary bypass and extracorporeal life support.
Industrial Applications & Lab Scenarios
- Cardiovascular device manufacturers – recirculation performance validation of dual-lumen cannulae per ISO 18193
- Third-party certification laboratories – traceable compliance testing for extracorporeal circulation devices
- R&D departments – optimization of dual-lumen cannula design to minimize recirculation
- Quality control teams – batch verification of cannulae used in CPB, ECLS and ECCO₂R systems
- Regulatory compliance teams – demonstration of safety and performance characteristics for market approval

Strategic Procurement Advantages & Global Support
- High-Efficiency Testing: Precision anatomical model enables fast, repeatable ISO 18193 Figure C.2 recirculation testing with clear visual results.
- Certified Manufacturing: ISO 9001/14001/45001 and CE certified production with ISO 17025 traceability ensures laboratory-grade reliability.
- Reliability & Support: Each unit undergoes rigorous factory calibration in our Dongguan facility and is backed by a full one-year warranty, on-site installation guidance, and lifetime technical engineering support.
Compliance & Regulatory Assurance
This equipment is manufactured in full accordance with ISO 18193 Figure C.2 for recirculation testing of dual-lumen cannulae used in extracorporeal circulation. It provides a standardized, reproducible test fixture essential for demonstrating compliance with international requirements for cannula safety and performance.
For detailed information on test procedures, please refer to the latest edition of ISO 18193. Regular visual inspection and gentle cleaning are recommended to maintain model clarity and dimensional accuracy.
Technical Inquiry & Expert Support
KingPo provides validated ISO 18193 Figure C.2 vascular models for cannula recirculation testing. Contact our medical engineering team for 1:1 anatomical scale configurations and setup support.
The Core Value We Deliver
- ISO 18193 Compliance: Exact 1:1 scale of SVC, RA, and IVC geometry.
- Optical Clarity: High-transparency design for clear flow visualization (ink/particles).
- Repeatable Results: Precision-machined to eliminate geometric variance.
- Lab Ready: Seamless integration with standard extracorporeal test circuits.
- Fast Response: Get a tailored technical recommendation within 24 hours.
Detail Display
ISO 18193 Figure C.2 Vascular Model FAQs
Related Products
Related News
ISO 80369-7 Figure C.2 Male Luer Lock Reference Connector Our 80369-7 compliant Figure C.2 male luer reference connectors are precision-engineered […]
Essential equipment for residual voltage testing under IEC 60601-1, IEC 60335-1, IEC 61010-1, IEC 62368-1 and similar safety standards. 1. […]
ISO 5356-1:2015 is the internationally recognized standard titled Anaesthetic and respiratory equipment — Conical connectors — Part 1: Cones and […]
You rely on the 6% taper angle when using iso 80369-7:2021 luer connectors. This special shape helps iso 80369-7 connectors […]











