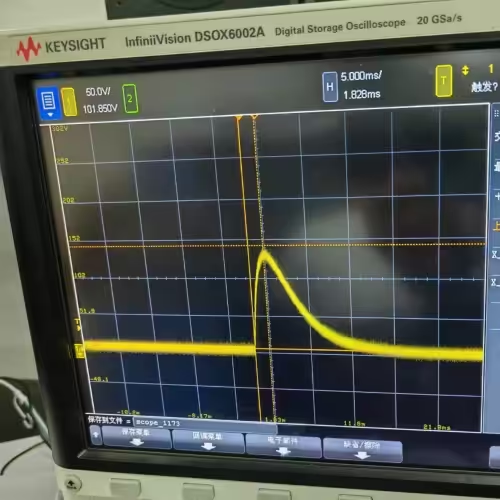
ISO 14708-1 clause 20.1, Test 1/2 Defibrillation Test Pulse Generator
KP-1050S Defibrillation Test Pulse Generator: All-in-one AIMD tester for Test 1 (damped sinusoidal 140/380V) & Test 2 (truncated exponential 140/270V, single/dual-phase). ISO 14117, ISO 14708-1 & GB 16174 compliant. Integrated solution vs separate units.
- All-in-One Dual-Mode Testing Integrates Test 1 (damped sinusoidal, 140/380 V) and Test 2 (truncated exponential, 140/270 V, single/dual-phase) in one compact unit — switch modes easily with the front-panel toggle.
- Intuitive Touchscreen Operation Color touchscreen for quick voltage setting, charge/discharge control, polarity selection, real-time capacitor voltage display, pulse counting, timer, and status monitoring — very user-friendly.
- Automated Compliance Sequences Automatically performs standard test sequences (3 positive pulses → 60 s pause → 1 negative pulse) with 1–999 programmable cycles, fully compliant with ISO 14117, ISO 14708-1, GB 16174.1-2024, EN 45502.
- High Safety & Reliable Design High-voltage warnings (>1000 V), IEC Class I grounding, external resistor network (KP-1050-1), robust build (≈10 kg) — safe and durable for medical device labs.
- Cost-Effective & Efficient Combines two test types into one affordable instrument with easy calibration and parameter memory — ideal for AIMD manufacturers, QA, and certification labs.
Defibrillation Test Pulse Generator Description
KP-1050S Defibrillation Test Pulse Generator – All-in-One AIMD Defibrillation Immunity Tester (clause 20.1 Test 1 & Test 2 Combined)
The KP-1050S Defibrillation Test Pulse Generator is a versatile, all-in-one compliance test instrument for assessing defibrillation immunity in active implantable medical devices (AIMDs) like pacemakers and ICDs. It generates both damped sinusoidal (Test 1) and truncated exponential (Test 2, single-phase or dual-phase) waveforms, fully compliant with GB 16174.1-2024 20.2, ISO 14117:2012, ISO 14708-1:2003, EN 45502-1:2015, and EN 45502-2-2:2008. Unlike separate testers (e.g.,Other competing products in the industry Implantable Test 1 for damped sinus and Test 2 for truncated exponential), the KP-1050S integrates both test modes via a simple front-panel switch, offering efficient workflow for labs and manufacturers.
The KP-1050S fromKINGPO LTD is a compact, touchscreen-operated pulse generator that simulates defibrillation shocks for AIMD safety testing. It supports both damped sinusoidal (Test 1) and truncated exponential waveforms (Test 2, selectable single/dual-phase), with automated sequences, polarity switching, and external resistor network (KP-1050-1). This integrated design provides a cost-effective alternative to separate testers like Other equipment in the industry Implantable series (Test 1 for damped sinus, Test 2 for monophasic/biphasic truncated exponential), streamlining compliance verification.
Key Features (Updated & Supplemented):
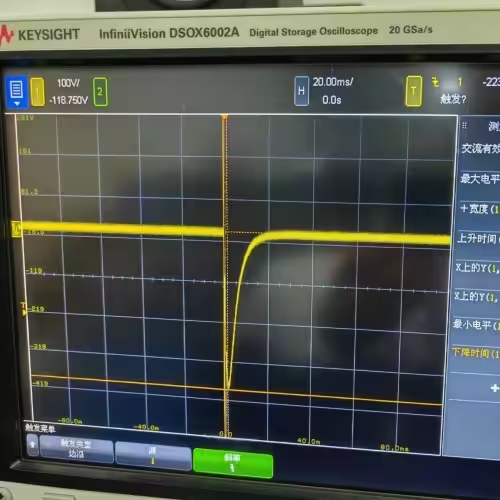
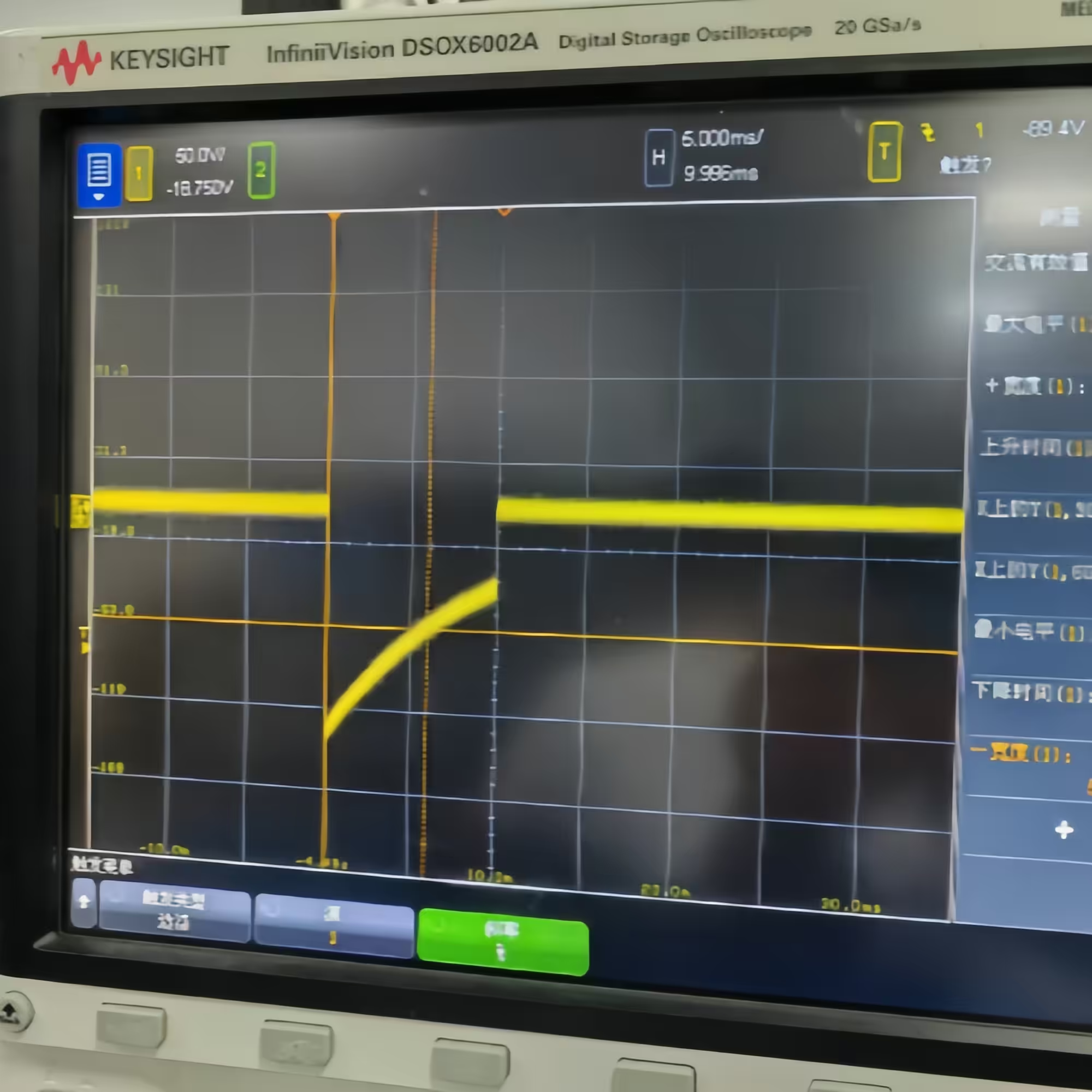
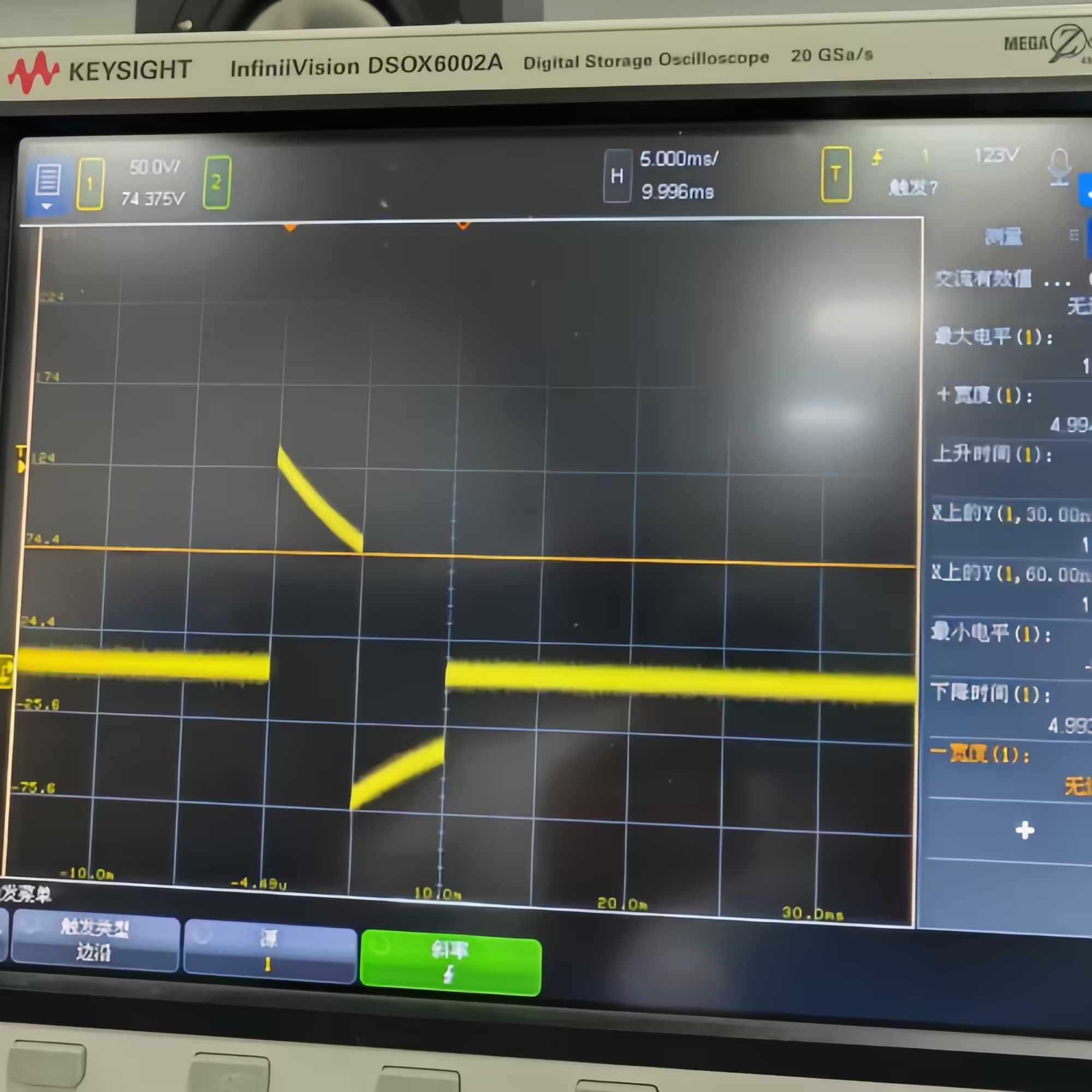
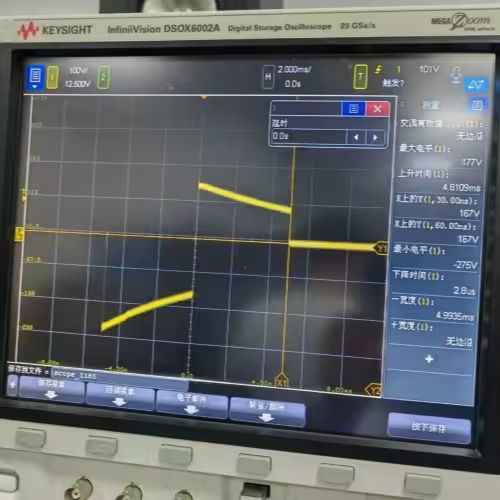
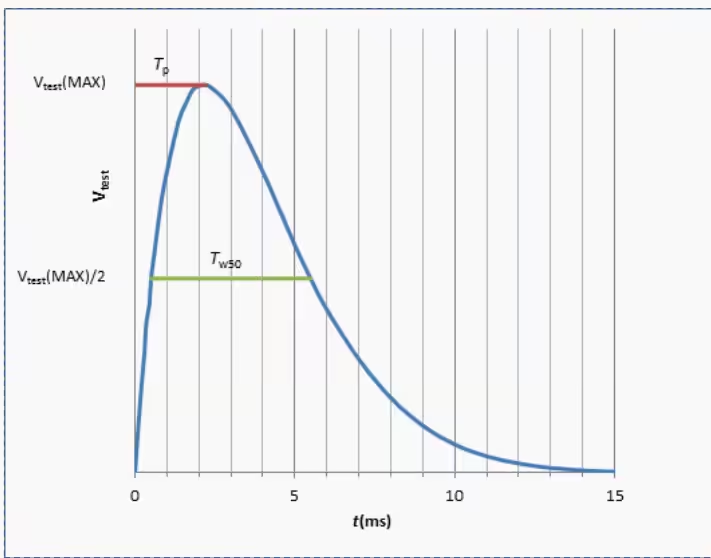
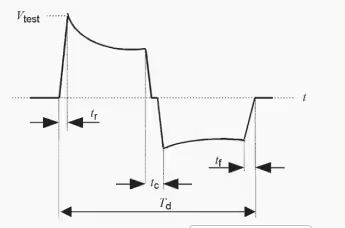
- Dual-mode operation: Test 1 (damped sinusoidal, 140V/380V ±5%/-0%, Tp 1.5–2.5 ms, Tw50 3–5.5 ms) and Test 2 (truncated exponential, 140V/270V ±5%/-0%, Td 9.5–10.5 ms, rise/fall 1–5 μs) in one unit
- Polarity: Freely selectable positive/negative; automated sequences (3 positive pulses at 20±2 s intervals, 60±2 s pause, then 1 negative pulse)
- Phase switching for Test 2: Single-phase or dual-phase (biphasic/monophasic)
- Touchscreen interface for voltage setting, charge/discharge control, single/dual pulse selection, timer, pulse counting (+/-), and real-time status (Charging, Ready, Discharging, Complete)
- Capacitor voltage monitoring (separate for Test 1 & Test 2) and calibration interface with positive/negative compensation
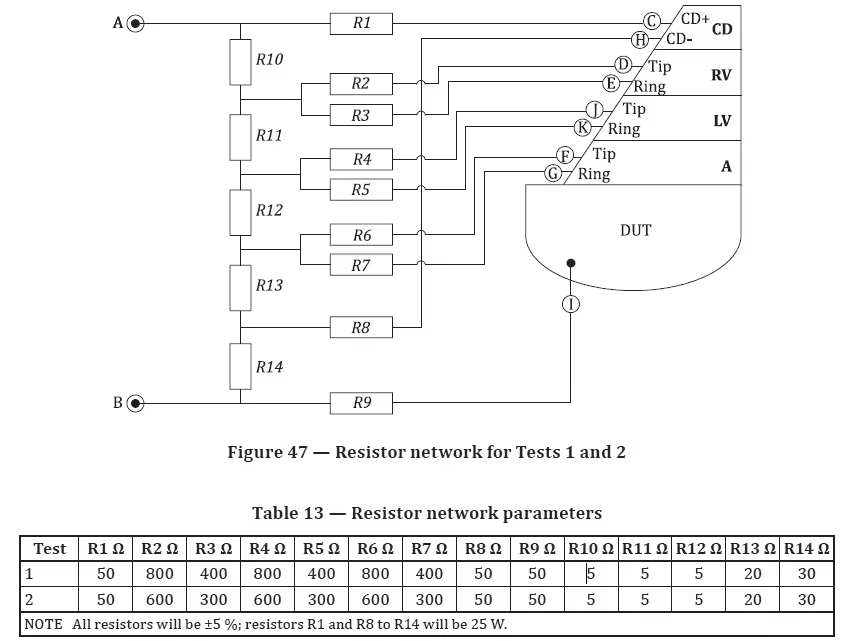
- External test network: KP-1050-1 resistor box (per ISO 14117 resistor parameters for Tests 1/2)
- Test count: 1–999 programmable
- Safety: High-voltage warnings (>1000V), IEC Class I grounding, interlock-like controls via power and discharge
- Dimensions: 480 × 460 × 200 mm; Weight: ~10 kg
- Power: AC 220/110V ±10%, 50/60Hz (max 300W)
- Compared to Compliance West MegaPulse Implantable Test 1 (damped sinus only, internal networks, 20 s recharge) and Test 2 (truncated exponential monophasic/biphasic, 140–270V), the KP-1050S combines both in one device with touchscreen convenience and external network flexibility.
Defibrillation Test Pulse Generator Technical
| Parameter | Specification |
|---|---|
| Applicable Standards | GB 16174.1-2024 20.2, ISO 14117:2012, ISO 14708-1:2003, EN 45502-1:2015, EN 45502-2-2:2008 |
| Test 1 Voltage | 140 V / 380 V (Tolerance: +5% / -0%) |
| Test 1 Circuit | RLC Circuit |
| Damped Sinusoidal Defibrillation Waveform | 1.5 ms < Tp < 2.5 ms; 3 ms < Tw50 < 5.5 ms |
| Output Polarity | Positive and negative polarity can be freely set |
| Test 1 Sequence | Send 3 positive voltage pulse sequences first (20 ± 2 s interval between each pulse), then after 60 ± 2 s interval, send one negative voltage pulse |
| Test 2 Voltage | 140 V / 270 V (Tolerance: +5% / -0%) |
| Phase Switching | Single-phase / dual-phase can be freely set |
| Single/Dual-phase Truncated Exponential Defibrillation Waveform | 9.5 ms < Td < 10.5 ms; 1 μs < tr < 5 μs; 1 μs < tf < 5 μs |
| Test 2 Sequence | Send 3 positive single-phase voltage pulse sequences first (20 ± 2 s interval between each pulse), then after 60 ± 2 s interval, send one negative voltage pulse |
| Evaluation Criteria | Confirm compliance by verifying whether active implantable medical devices are not permanently affected after completing the full Test 1 and Test 2 procedures, and whether they can be reprogrammed to restore settings |
| External Test Network | Test Network Box: KP-1050-1 (or KP-10505-1 variant) |
| Test Count | 1 – 999 times |
| Input Power | AC 220/110 V ±10%, 50 Hz / 60 Hz (max power approx. 300 W) |
| Dimensions | 480 × 460 × 200 mm |
| Weight | Approx. 10 kg |
ISO 14708-1 clause 20.1, Test 1/2 Defibrillation Test Pulse Generator Applications
- EN 45502-1:2015, clause 20.1, Test 1, Figures 1, 2 and 3; 140V peak.
- ISO 14117:2012, clause 6.2.2, Test 1; Figures 45 and 46; 380V peak.
- ISO14708-1:2014, Part 1; 140V peak.
- EN 45502-2-2:2008, clause 20.2; 380V peak.
- EN 45502-2-1:2003, 140V peak.
- EN 45502-2-2-3:2010, 140V peak.
KP1050S Defibrillation Test Pulse Generator CALIBRATION CERTIFICATE
KP1050S Defibrillation Test Pulse Generator User Manual
ISO 14708-1 clause 20.1, Test 1/2 Defibrillation Test Pulse Generator Detail Display
Implantable Surge Tester FAQS
Related Products
Related News
Understanding the ESU Analyzer: A Comprehensive Guide In the world of medical equipment, precision and safety are paramount. One device […]
The Defibrillation Immunity Testing Requirements for Active Implantable Medical Devices (AIMDs): Standards Breakdown, Visual Insights, and Recommendation for the KP-1050S […]
Shan Chao 1 , Qiang Xiaolong 2 , Liu jiming 3 , Zhang Chao 3 . (1. Heilongjiang Institute for […]
Abstract Thermage, a non-invasive radiofrequency (RF) skin tightening technology, is widely used in medical aesthetics. With operating frequencies increasing to […]