
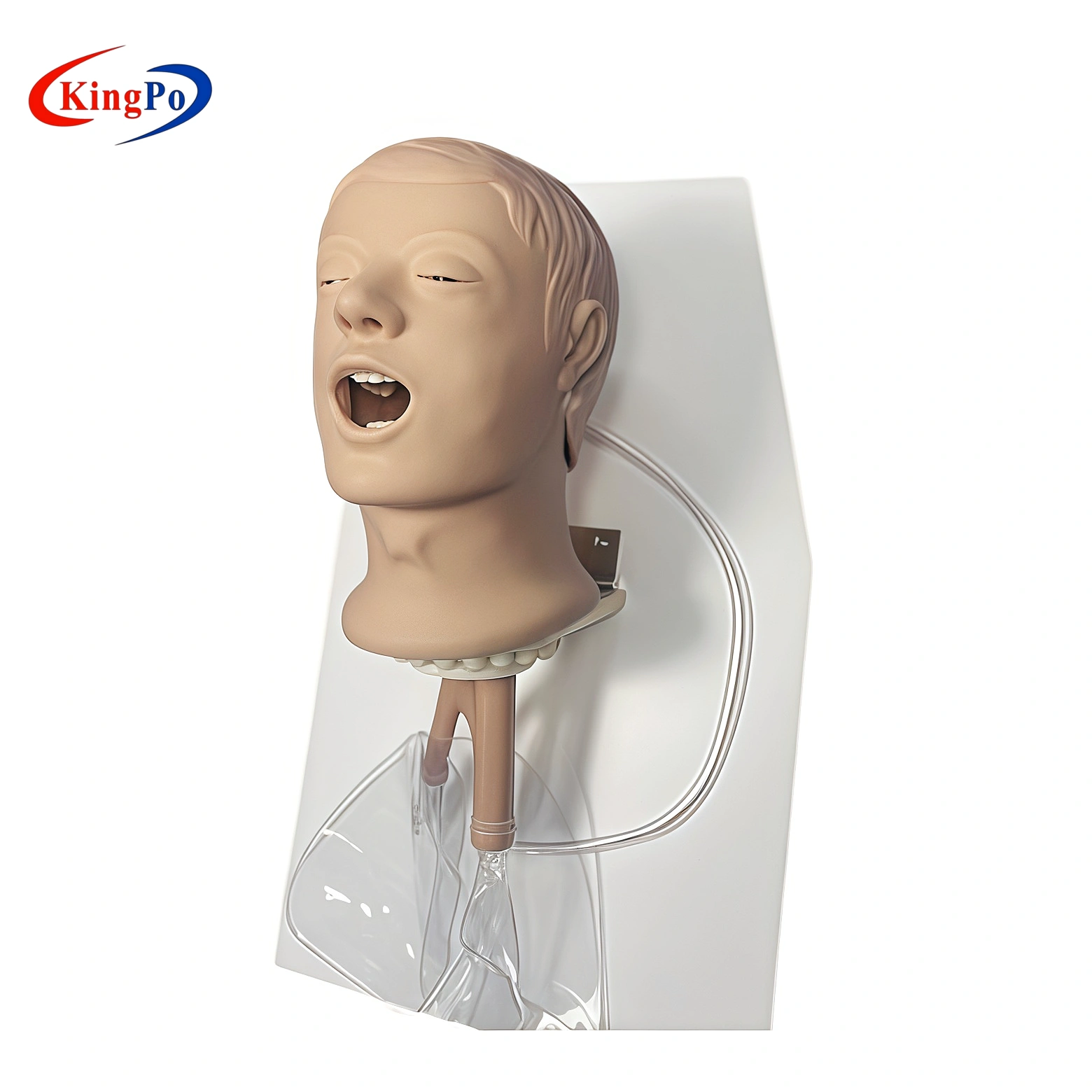



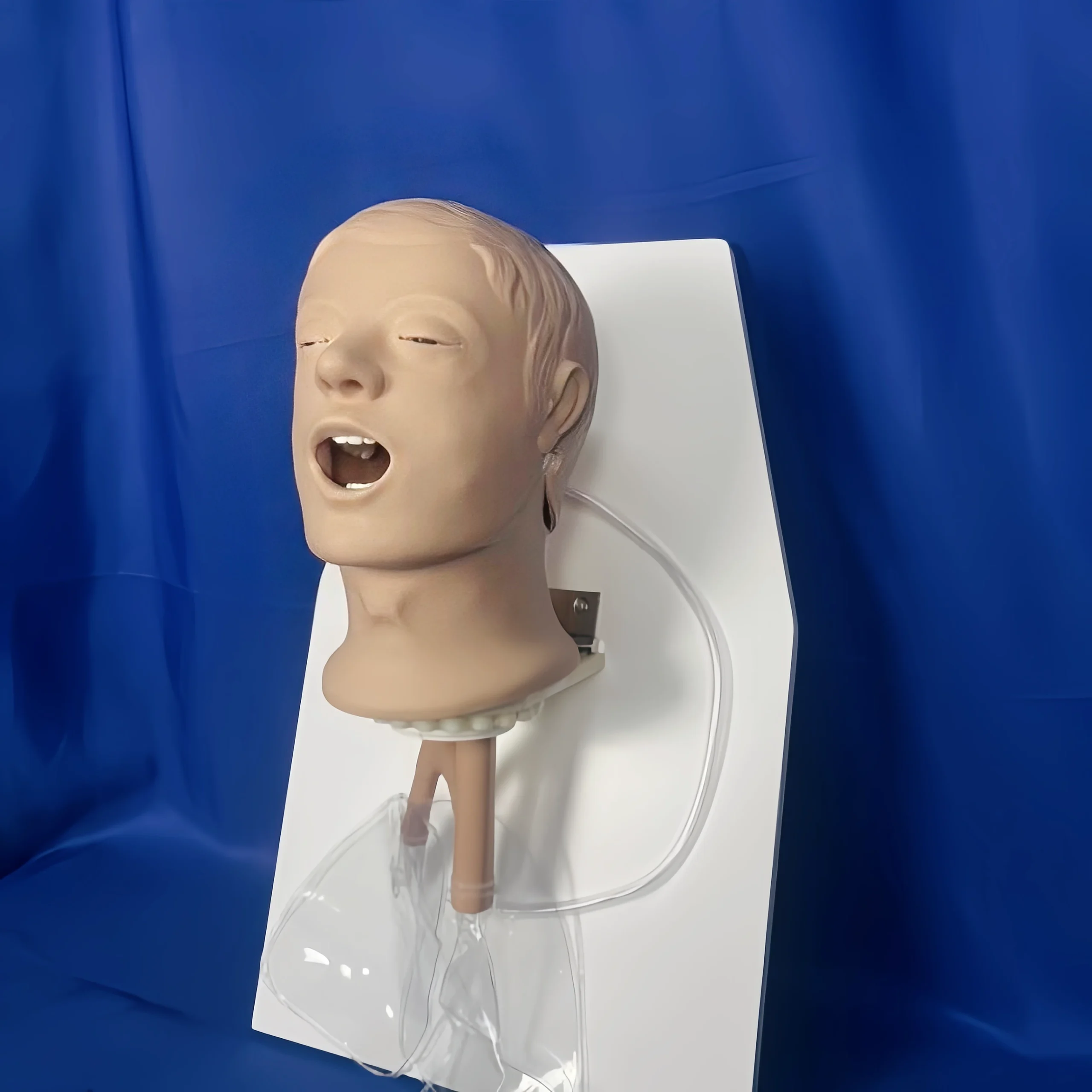
ISO 9360-2 Figure 1 Simulated Patient Head | Passive HME Testing Model
The ISO 9360-2 Figure 1 Simulated Patient Head is a high-precision plastic anatomical model designed for performance testing of passive heat and moisture exchangers (HMEs) intended for tracheostomized patients.
- Standard Compliance: ISO 9360-2 Figure 1 – exact simulated patient head for passive HME testing
- Material: High-quality durable medical-grade plastic
- Application: Testing of passive HMEs for tracheostomized patients (tidal volume ≥250 ml)
- Anatomical Accuracy: Precise replication of human head and upper airway geometry per ISO 9360-2
- Compatibility: Designed for HME moisture loss and pressure drop measurement
- Warranty: Full 1-year manufacturer warranty
- MOQ: 1 /
- Delivery Period: 20 working days
ISO 9360-2 Figure 1 Simulated Patient Head Description
Plastic Material Simulated Patient Head with ISO 9360-2-figure 1 Standard
The ISO 9360-2 Figure 1 Simulated Patient Head | Passive HME Testing Model is a professional anatomical simulation device manufactured by KingPo. It is specifically designed to replicate the human head and upper airway for performance testing of passive heat and moisture exchangers (HMEs) intended for tracheostomized patients, in full accordance with ISO 9360-2 Figure 1.
Technical Parameters
| Parameter | Specification | Remark / Notes |
|---|---|---|
| Standard | ISO 9360-2 Figure 1 | Exact anatomical simulation for passive HME performance testing (moisture loss and pressure drop) |
| Model | KP-MNT10 | Precision test fixture for tracheostomized patient airway simulation |
| Material | Medical-grade engineering plastic | Non-porous, corrosion-resistant, smooth surface finish for repeated cleaning and long-term dimensional stability |
| Anatomical Replication | Full compliance with ISO 9360-2 Figure 1 | Precise replication of human head geometry and upper airway channel |
| Internal Airway Design | Controlled internal volume and geometry | Ensures accurate gas flow path and repeatable moisture exchange measurements |
| Mounting Base | Stable, vibration-free mounting platform | Compatible with standard laboratory test rigs and breathing circuits |
| Surface Properties | Smooth, non-porous | Facilitates easy cleaning and prevents residue accumulation |
| Certification | ISO 17025 traceable | Supports laboratory accreditation and audit requirements |
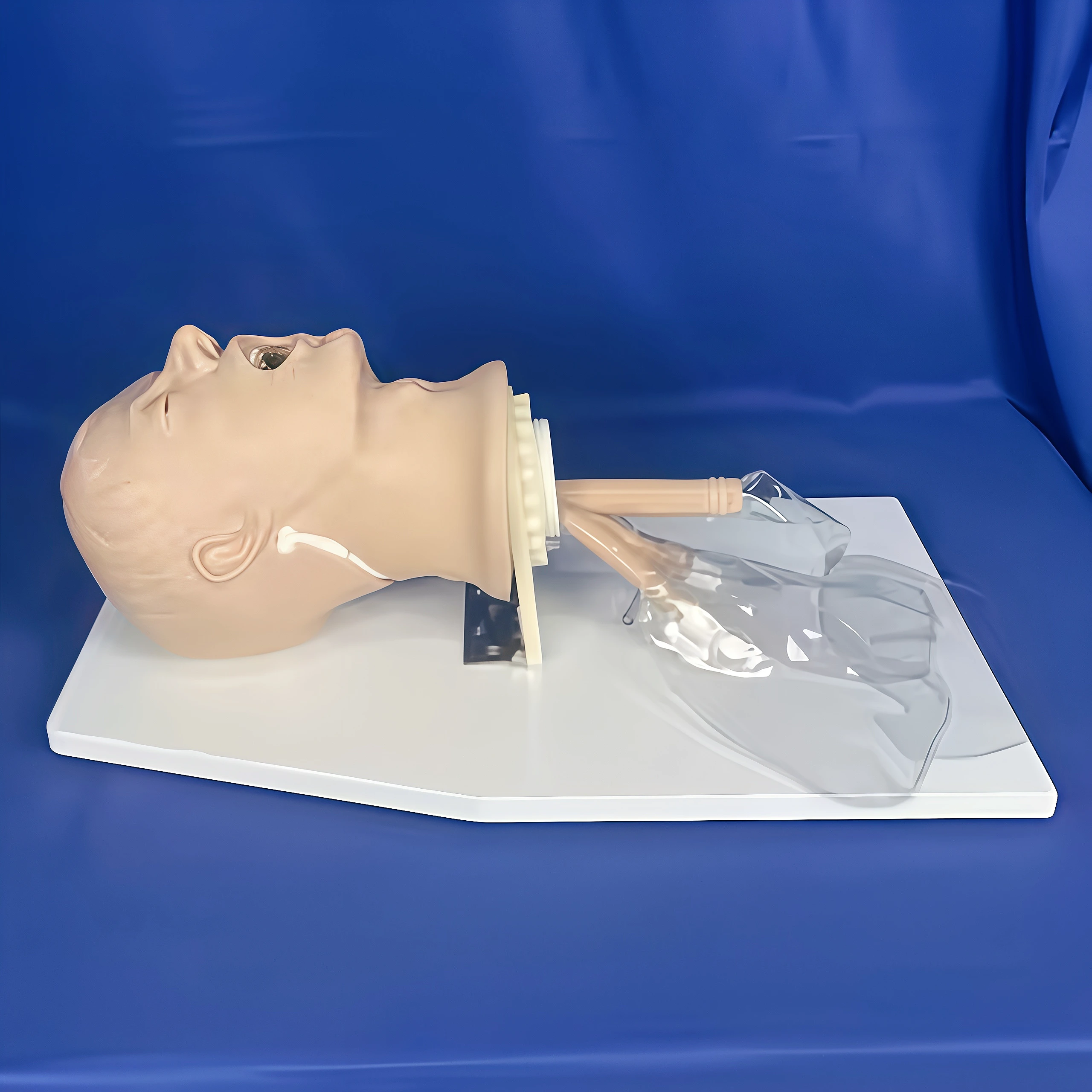
The Simulated Patient Head is precision-molded from high-grade engineering plastic that exactly replicates the geometry and surface characteristics of the human head and upper airway as defined in ISO 9360-2 Figure 1. The internal airway channel is engineered with precise dimensional tolerances to ensure accurate gas flow, moisture loss, and pressure drop measurements.
All external surfaces are smooth and non-porous for easy cleaning, while the stable mounting base provides vibration-free attachment to test rigs. Realistic anatomical landmarks allow correct placement of HMEs and breathing circuits, eliminating variability in test results.
Testing Principle
The device simulates the upper airway of a tracheostomized patient. A passive HME is connected to the head’s patient port while controlled humidified and heated gas flows through the system. The HME’s ability to retain and return moisture and heat from exhaled gas is measured according to the test methods in ISO 9360-2 (moisture loss and pressure drop).
Common Error Mitigation
To ensure repeatable and accurate results, always confirm the HME is fully seated on the patient port without gaps and that test gas flow matches the minimum tidal volume requirement (≥250 ml). Regular gentle cleaning of the airway channel prevents residue buildup that could affect moisture exchange performance.
-
- SO 9360-2 Figure 1 Simulated Patient Head
Primary Use Cases & Business Value
The Simulated Patient Head is used by medical device manufacturers and certification laboratories to evaluate the humidification performance of passive HMEs for tracheostomized patients. It enables accurate, standardized testing that ensures patient safety, regulatory compliance, and reliable respiratory support in clinical settings.
Industrial Applications & Lab Scenarios
- Respiratory device manufacturers – performance validation of passive HMEs per ISO 9360-2
- Medical training and simulation centers – realistic anatomical training for tracheostomy care
- Third-party certification laboratories – traceable compliance testing of heat and moisture exchangers
- R&D departments – development and optimization of new HME designs for tracheostomized patients
- Quality control teams – batch verification of HMEs used in anesthesia and critical care
Strategic Procurement Advantages & Global Support
- High-Efficiency Testing: Precision anatomical model enables fast, repeatable ISO 9360-2 Figure 1 testing with minimal setup time.
- Certified Manufacturing: ISO 9001/14001/45001 and CE certified production with ISO 17025 traceability ensures laboratory-grade reliability.
- Reliability & Support: Each unit undergoes rigorous factory calibration in our Dongguan facility and is backed by a full one-year warranty, on-site installation guidance, and lifetime technical engineering support.
Compliance & Regulatory Assurance
This equipment is manufactured in full accordance with ISO 9360-2 Figure 1 for testing passive heat and moisture exchangers used with tracheostomized patients. It provides a standardized, reproducible test fixture essential for demonstrating compliance with international respiratory equipment safety and performance requirements.
For detailed information on testing procedures, please refer to the latest edition of ISO 9360-2. Regular visual inspection and gentle cleaning are recommended to maintain dimensional accuracy.
Technical Inquiry for Simulated Patient Head
If you require the ISO 9360-2 Figure 1 Simulated Patient Head or custom configurations, our engineering team can provide technical support and configuration recommendations.
The Core Value We Deliver to You
- Exact replication of ISO 9360-2 Figure 1 geometry for accurate HME testing
- Durable medical-grade plastic construction with long service life
- 1-year warranty and ISO 17025 traceable calibration support
- Simple, stable mounting for repeatable laboratory results
- Ideal for both product development and regulatory compliance testing
Tell us your HME type or test requirements — our engineering team will reply with tailored recommendations within 24 hours.
Detail Display
Simulated Patient Head ISO 9360-2 FAQs
Related Products
Related News
NE Contact Impedance Tester for Contact Resistance Accuracy NE Contact Impedance Tester In the world of electrical testing equipment, the […]
Conflict of interest disclosure: This is a first‑party, engineering‑style review of the Kingpo KP‑8850 ESU analyzer. We aim to be […]











