



KP-249 Interference Coupling Network/HFCN Test Fixture
The KP-249 High Frequency Surgical Equipment Interference Coupling Network is a precision testing device compliant with IEC 80601-2-26, IEC 60601-2-27, and IEC 80601-2-49 standards. It facilitates accurate interference and protective measurements for high-frequency surgical devices, ensuring enhanced patient safety and regulatory compliance in medical environments.
- Versatile Standard Compliance: Supports multiple IEC standards for emissions, immunity, and protective measurements, allowing flexible testing across various scenarios and reducing the need for multiple devices.
- Advanced Shielding and Resistor Design: Features a metal shielded enclosure and high-power non-inductive resistors for precise patient impedance simulation, minimizing interference and ensuring reliable, accurate results.
- Comprehensive ECG Integration: Compatible with 10-lead ECG and multi-parameter monitors, enabling thorough monitoring and testing of patient-connected equipment to enhance safety protocols.
- User-Friendly Switchable Modes: Includes easy-to-toggle switches for shielded/non-shielded and conductive/non-conductive configurations, streamlining setup and improving efficiency in compliance testing workflows.
HFCN Test Fixture Description
Discover the KP-249 High Frequency Surgical Equipment Interference Coupling Network, a specialized testing device designed for IEC 80601-2-26, IEC 60601-2-27, and IEC 80601-2-49 standards. This robust network ensures accurate protective measurements for high-frequency surgical devices, featuring metal shielding, high-power non-inductive resistors, and support for 10-lead ECG monitoring. Ideal for medical device manufacturers seeking reliable compliance testing solutions to enhance patient safety and equipment performance.
Focus Keyword
KP-249 Interference Coupling Network
Category Primary Title
Medical Device Compliance Testing Equipment
Category Short Introduction
Explore our range of advanced medical device compliance testing equipment, engineered to meet stringent international standards like IEC 60601 series. These tools facilitate precise interference and protective measurements for high-frequency surgical devices, ensuring regulatory adherence and optimal safety in healthcare environments.
Features
- Metal Shielded Enclosure: Provides superior internal shielding to minimize external interference during testing.
- Built-in High-Power Non-Inductive Resistors: Enables stable and accurate simulations of patient impedance without inductive effects.
- 10-Lead ECG Support: Compatible with multi-parameter patient monitors for comprehensive 10-channel connections.
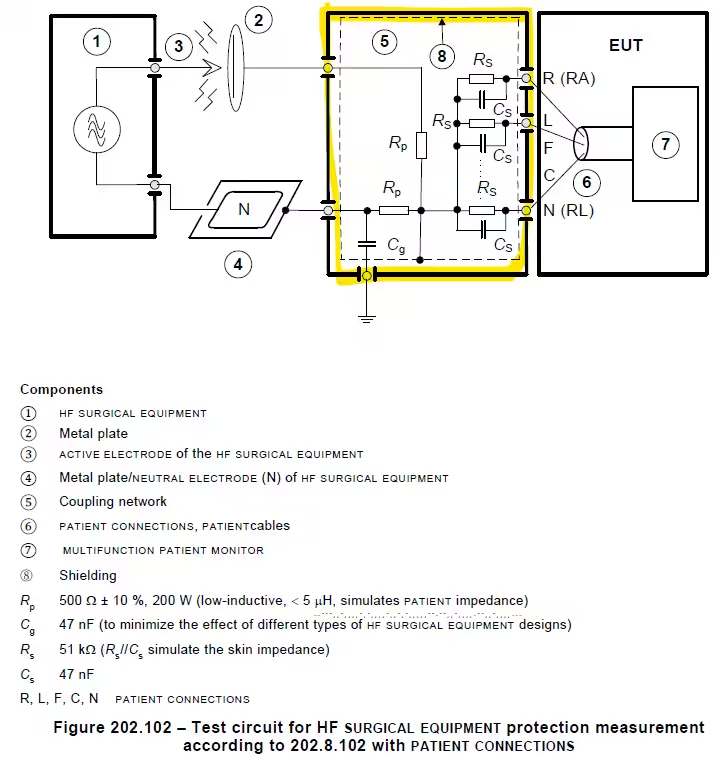
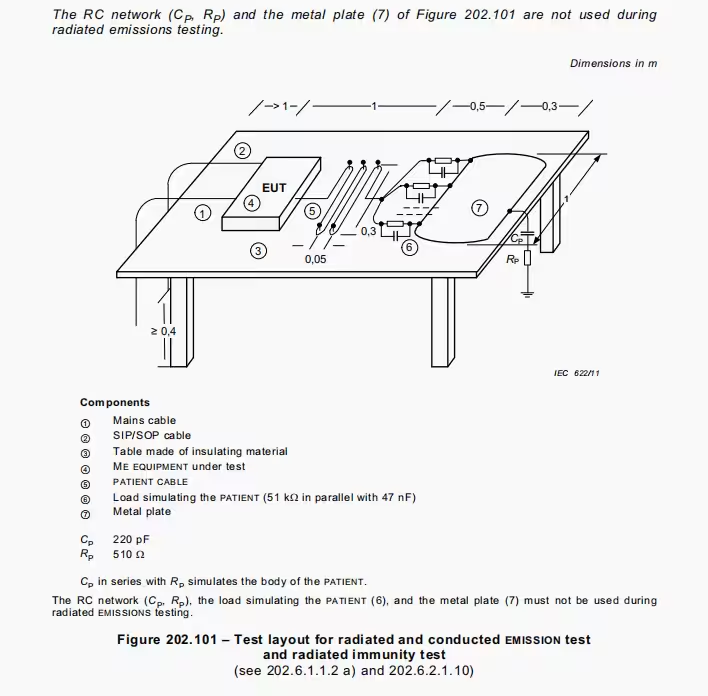
- Versatile Standard Compatibility: Supports testing under IEC 60601-2-26 (radiation and conducted emissions), IEC 60601-2-27 (conducted emissions and radiation immunity), and IEC 80601-2-49 (protective measurements with or without patient connections).
- Switchable Shielding and Conductivity Options: Easily toggle between shielded/non-shielded and conductive/non-conductive modes for diverse test scenarios.
- Grounding and Interface Connections: Includes yellow ground wire for secure earthing, RE&CE interfaces for patient cables or copper foils, and RC networks for simulating patient body impedance.
Product Series Title
KP-249 Series: Advanced Interference Coupling Networks for Surgical Equipment
Product Series Content Introduction
The KP-249 Series represents a cutting-edge line of interference coupling networks tailored for high-frequency surgical equipment testing. Built to comply with key medical standards such as IEC 60601-2-26, IEC 60601-2-27, and IEC 80601-2-49, this series offers versatile configurations for protective measurements, emissions testing, and immunity assessments. Whether handling patient-connected devices or non-conductive applications, the KP-249 ensures precise results through its shielded design, multi-lead ECG integration, and customizable switches. Perfect for laboratories and manufacturers prioritizing accuracy, safety, and efficiency in medical device validation.
- Elaborated design offers convenience and safety
- Dual module design supports different standard requirements
- Built-in impedance 51k/47nF for skin impedance simulation
- Simulate 1mV ECG signal for electrosurgical interference suppression (ESIS)
- Power rating >300W and resistor inductance <5μH
- Able to use BB-C1 breakout box bundled with SECG 4.0 and MECG 2.0
KP-249 High Frequency Surgical Equipment Interference Coupling Network Technical Specifications
Unlock the full potential of the KP-249 High Frequency Surgical Equipment Interference Coupling Network with these detailed technical specifications. Engineered for precision in medical device testing, this device adheres to international standards, offering robust features for interference coupling and protective measurements.
| Specification | Details |
|---|---|
| Compliance Standards | IEC 80601-2-26 (Radiation and Conducted Emissions), IEC 60601-2-27 (Conducted Emissions and Radiation Immunity), IEC 80601-2-49 (High-Frequency Surgical Equipment Protective Measurements) |
| Enclosure Design | Internal metal shielded box for enhanced electromagnetic interference protection |
| Resistors | Built-in high-power non-inductive resistors for accurate patient impedance simulation |
| ECG Compatibility | Supports 10-lead ECG monitoring with multi-parameter patient monitor connections |
| Switch Options | – Shield switch: Toggle between shielded and non-shielded modes – Conductive switch: Toggle between conductive and non-conductive modes |
| Interfaces and Connections | – Surgical electrode and neutral electrode ports – 10-channel rear connections for patient monitors – Yellow ground wire interface for secure earthing – RE&CE interfaces for patient cables or copper foil (patient impedance simulation) – RC network connection for serial patient body simulation on metal plates |
| Testing Modes | – Patient-connected protective measurements (IEC 80601-2-49) – Non-conductive application parts testing (IEC 80601-2-49) – Conducted emissions and radiation immunity (IEC 60601-2-27) – Radiation and conducted emissions (IEC 80601-2-26) |
| Applications | Suitable for medical laboratories, device manufacturers, and compliance testing facilities focusing on high-frequency surgical equipment safety and performance |
Conversion Module
Ready to enhance your medical device testing capabilities? Request a quote for the KP-249 Interference Coupling Network today and ensure your equipment meets global standards. Contact our sales team
KP-249 High Frequency Surgical Equipment Interference Coupling Network Applications
The KP-249 Interference Coupling Network is essential for a variety of medical testing applications, including protective measurements, emissions compliance, and immunity assessments in high-frequency surgical environments. Below are key applications:
- Protective Measurements for Patient-Connected Devices (IEC 80601-2-49): Simulates real-world interference to verify safety in surgical procedures involving direct patient contact.
- Non-Conductive Application Testing (IEC 80601-2-49): Evaluates equipment with non-conductive parts, using copper foil simulations for accurate impedance checks.
- Conducted Emissions and Radiation Immunity (IEC 60601-2-27): Tests patient cable connections and RC networks to ensure resilience against electromagnetic disturbances.
- Radiation and Conducted Emissions (IEC 80601-2-26): Measures emissions from surgical devices, supporting metal plate integrations for thorough compliance validation.