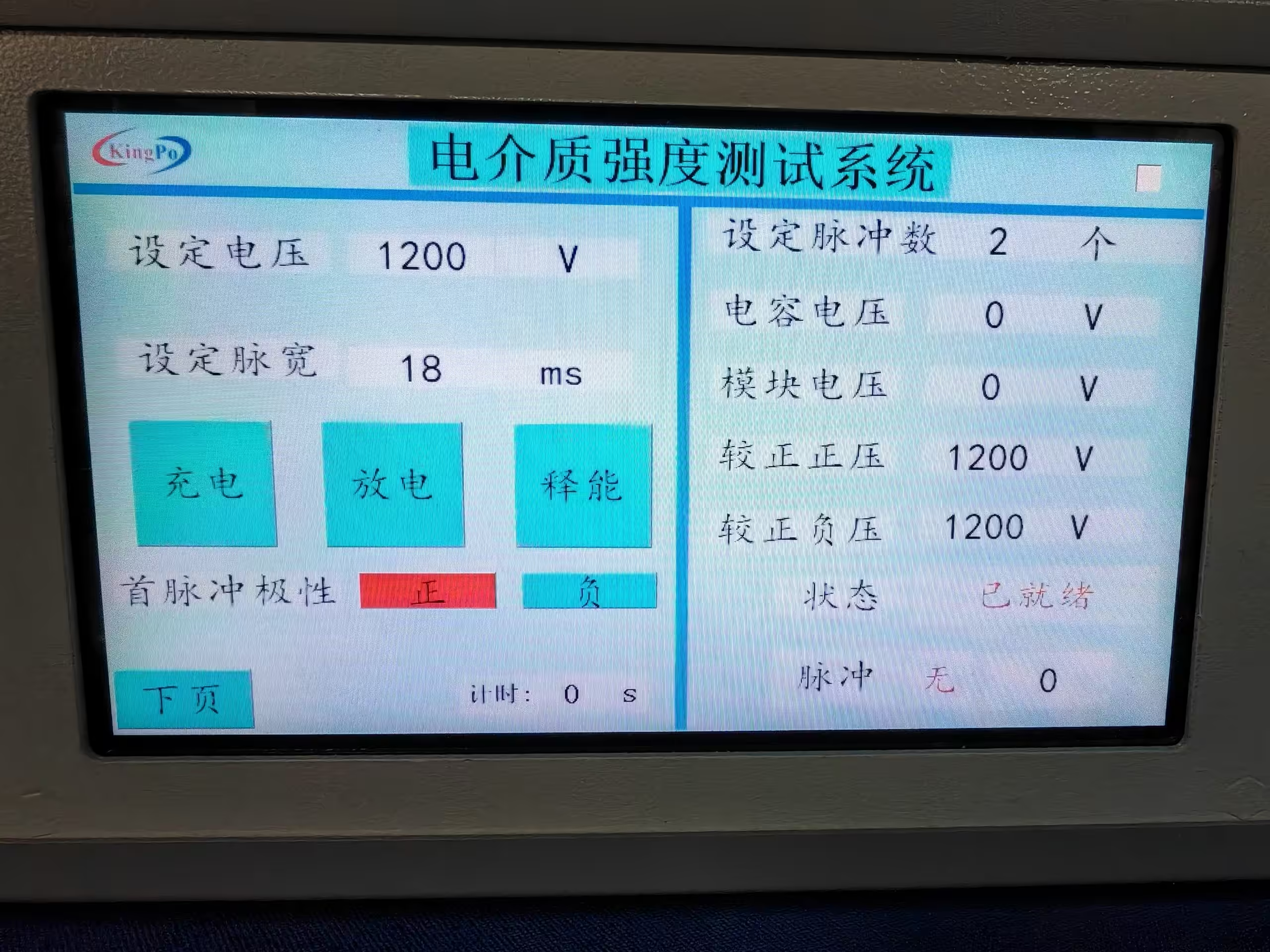
KP-1230S Dielectric Strength Tester Compliant with ISO 27186:2020 Annex C
The KP-1230S Dielectric Strength Testing System employs advanced high-voltage pulse generation technology to precisely reproduce the truncated exponential defibrillation waveform required by the standards. The system features a compact design, user-friendly operation, high-precision waveform monitoring, and data logging capabilities, supporting multiple test modes to ensure repeatable and accurate test results.
MOQ: 1
Price: To be quoted
Standard Packaging: 300*400*1500mm
Delivery Period: 15day
Payment Method: T/T
Supply Capacity: 100
Dielectric Strength Tester Product Overview
The KP-1230S Dielectric Strength Tester, developed and manufactured by KingPo Technology Development Limited, represents a specialized high-precision testing instrument designed specifically for evaluating the defibrillation resistance of quadripolar connector systems in active implantable medical devices. Compliant with stringent international standards including ISO 27186:2020 (Annex C) and YY/T 0972-2016 (Appendix C), this tester ensures that critical components in devices such as pacemakers, implantable cardioverter defibrillators (ICDs), and cardiac resynchronization therapy (CRT) devices can withstand high-voltage defibrillation pulses without permanent damage or loss of reprogrammability.
In clinical environments where patients rely on these life-sustaining implants, external defibrillation or internal shock delivery can pose risks to device integrity. The KP-1230S addresses this challenge by simulating real-world truncated exponential defibrillation waveforms, allowing manufacturers, testing laboratories, and quality control teams to verify connector reliability under extreme electrical stress conditions.
Core Technical Features and Waveform Precision
At the heart of the KP-1230S lies its advanced high-voltage pulse generation technology, which accurately reproduces the monophasic truncated exponential waveform mandated by relevant standards.
Key waveform parameters include:
- Peak Voltage: 1.53 kV (standard configuration, with adjustable range up to 2 kV)
- Pulse Duration: 18 ms
- Rise Time: 1.4 μs
- Voltage Rate of Change (dV/dt): 1.08 kV/μs
- Decay Requirement: >50% of peak voltage remaining after 18 ms
This waveform closely mimics the output of clinical defibrillators, delivering controlled high-energy pulses to the tested connector system while simulating human body impedance through standardized test loads. The system’s high-precision pulse control ensures repeatable test results, critical for regulatory compliance and design validation.
The tester supports comprehensive evaluation of whether the quadripolar connector experiences permanent structural or functional damage after exposure to such pulses, and whether device parameters can be successfully restored via reprogramming using an external programmer.
Safety, Usability, and Operational Design
Safety remains paramount in high-voltage testing. The KP-1230S incorporates multiple protection layers, including high-voltage isolation, automatic discharge circuits upon test completion or interruption, and an emergency stop function. These features minimize risks to operators while maintaining test accuracy.
The instrument adopts a compact desktop design, making it suitable for laboratory environments with limited space. It operates on standard AC 220V ±10%, 50/60 Hz power, with environmental tolerances of 10–40°C operating temperature and <80% relative humidity.
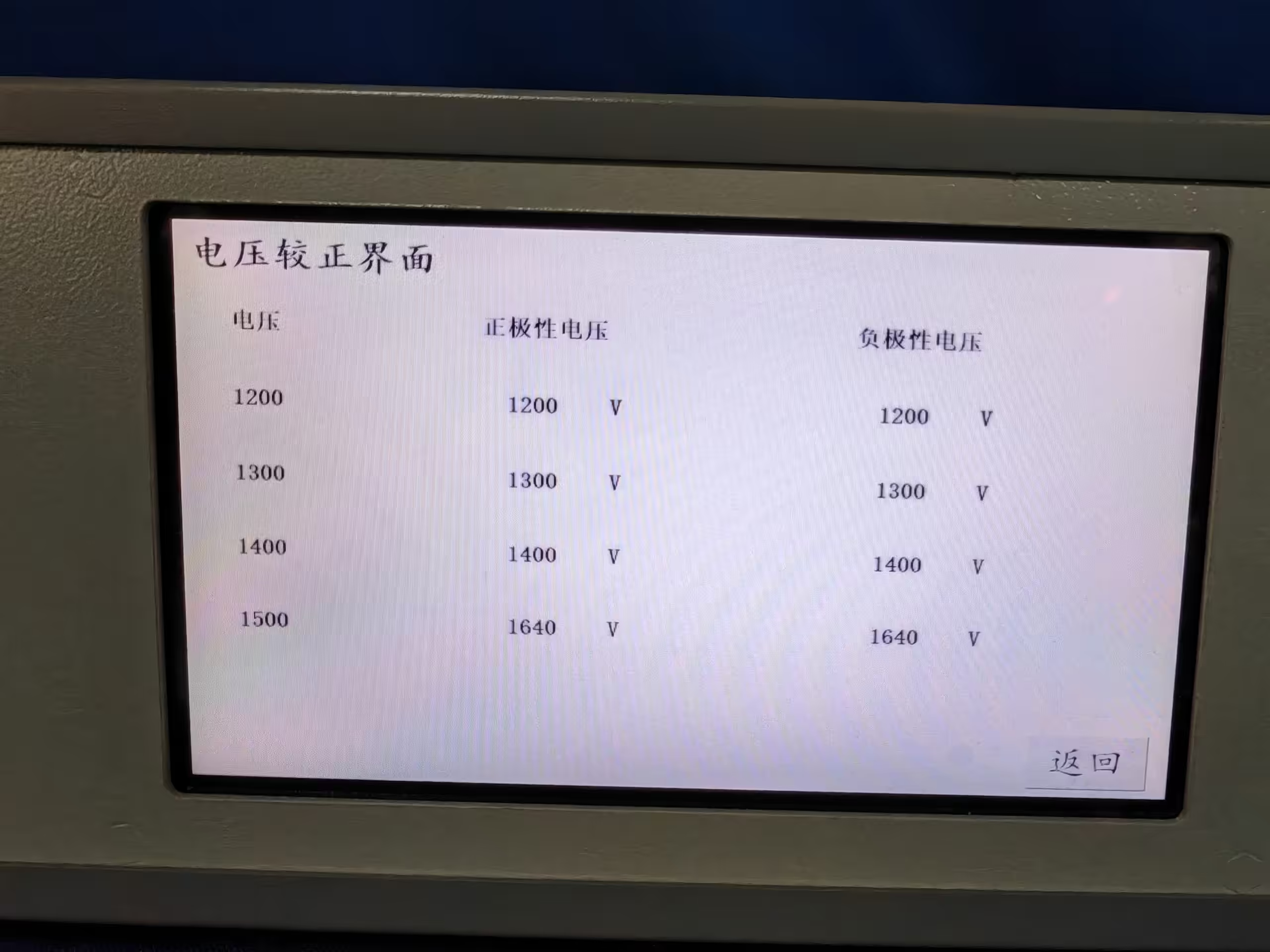
User-friendly operation is enhanced by intuitive controls, waveform monitoring capabilities, and data logging functions that facilitate detailed post-test analysis and report generation. Annual calibration is recommended to maintain long-term waveform accuracy and compliance.
Primary Applications and Industry Value
The KP-1230S serves multiple critical roles across the medical device lifecycle:
- R&D and Design Validation — Engineers use it to assess and optimize connector designs for defibrillation resilience, ensuring product reliability before clinical trials.
- Compliance Certification — Third-party testing labs rely on the system to generate standard-compliant reports for regulatory submissions (e.g., ISO 27186, Chinese YY standards).
- Production Quality Control — Manufacturers implement end-of-line or sampling tests to confirm batch consistency and prevent field failures.
- Research and Simulation — Academic and clinical research teams study defibrillation effects on implantable devices to advance patient safety technologies.
By confirming that devices remain functional after simulated defibrillation events, the KP-1230S directly contributes to protecting patients during emergency cardiac interventions.
Conclusion: Reliable Partner for Advanced Medical Testing
As a flagship product in KingPo’s medical test equipment portfolio, the KP-1230S Dielectric Strength Tester combines cutting-edge waveform simulation, robust safety features, and strict standard adherence into a compact, high-performance solution. Whether for implantable cardiac device manufacturers, certification bodies, or specialized research institutions, this tester provides the precision and confidence needed to meet evolving global regulatory requirements and uphold the highest standards of patient safety in active implantable medical technologies.
Dielectric Strength Tester, Defibrillation Waveform Testing, ISO 27186, YY/T 0972, Quadripolar Connector Testing, Implantable Cardiac Device Testing
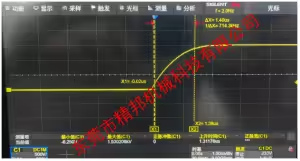
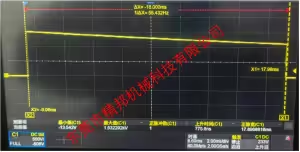
Dielectric Strength Tester Measured Waveform Data (Monophasic Truncated Exponential Wave)
| Parameter | Specification Value | Description |
|---|---|---|
| Test Signal Rise Time | 1.4 μs | Fast rise time to simulate real defibrillation shocks |
| dV/dt | 1.08 kV/μs | Voltage rate of change |
| Peak Voltage | 1.53 kV | Maximum output voltage |
| Pulse Duration | 18 ms | Total pulse width |
| Peak Voltage Decay Requirement | >50% of peak value after 18 ms | Complies with truncated exponential waveform decay characteristics |
Dielectric Strength Tester
If you need a quotation, demonstration, or customization services for the KP-1230S Dielectric Strength Tester, please contact us. We are committed to providing high-quality medical device testing solutions!
Application Scenarios
The KP-1230S Dielectric Strength Testing System is widely used in the following areas:
- Medical Device R&D and Validation: Defibrillation resistance testing of connector systems in implantable pacemakers, ICDs, and CRT devices to ensure compliance with safety standards before market release.
- Third-Party Testing Laboratories: Providing compliance certification testing services for manufacturers, supporting ISO 27186 and YY/T 0972 standard reports.
- Quality Control and Production Testing: End-of-line sampling testing to verify dielectric strength and reprogramming recovery of batch products.
- Clinical Simulation Research: Studying the effects of defibrillation shocks on implantable devices to optimize designs and improve patient safety.
In clinical practice, patients may receive external or internal defibrillation therapy. This system helps ensure that implanted devices will not suffer permanent damage under such high-voltage conditions, thereby protecting patient lives.
Other Technical Specifications
- Output Waveform Type: Monophasic Truncated Exponential Waveform
- Voltage Range: Adjustable, up to 2 kV (standard configuration 1.53 kV)
- Pulse Energy: Simulates clinical defibrillator output
- Load Adaptability: Supports standard test loads simulating human body impedance
- Power Requirements: AC 220V ±10%, 50/60 Hz
- Dimensions and Weight: Compact desktop design (specific dimensions vary by model)
- Environmental Requirements: Operating temperature 10-40°C, humidity <80%
These parameters ensure that the system output waveform fully meets the standard requirements for defibrillation protection testing.
Dielectric Strength Tester Measured waveform data:
Dielectric Strength Tester
FAQ (Frequently Asked Questions)
Related Products
Related News
Understanding the ESU Analyzer: A Comprehensive Guide In the world of medical equipment, precision and safety are paramount. One device […]
NE Contact Impedance Tester for Contact Resistance Accuracy NE Contact Impedance Tester In the world of electrical testing equipment, the […]
Dynamic Compensation Implementation for High-Frequency Electrosurgical Unit Testing Using High-Frequency LCR or Network Analyzers Above MHz Shan Chao1, Qiang Xiaolong2, […]
Enhancing Electrical Safety with IEC 60309 Compliance Gauges 39 I see electrical safety as a top priority in every industrial […]