
You use the ISO 18193 Figure C.2 vascular model to achieve accurate Dual Lumen Cannula Recirculation Testing. This model gives you a clear anatomical simulation that meets every requirement of the standard. You ensure compliance when you test dual lumen cannula devices because the model replicates the necessary vascular regions. Manufacturers, labs, and R&D teams rely on this solution for compliance and reliable testing results. You gain superior visualization and anatomical accuracy with this standard-compliant model.
Key Takeaways
-
The ISO 18193 Figure C.2 vascular model accurately simulates human vascular anatomy, essential for testing dual-lumen cannula devices.
-
Using this model ensures compliance with ISO standards, providing reliable data for cardiac diagnosis and treatment.
-
The model’s transparent design allows for clear observation of flow patterns, enhancing the accuracy of recirculation testing.
-
Regular calibration and precise manufacturing tolerances of the model lead to consistent and repeatable testing results.
-
Incorporating ink or particles during testing helps visualize flow dynamics, improving the understanding of device performance.
-
The model supports quality control and product development by identifying issues early and guiding design improvements.
-
Technical support and training are available to help users maximize the model’s effectiveness in testing and compliance.
-
Choosing the ISO 18193 Figure C.2 vascular model streamlines the certification process, saving time and resources in regulatory approvals.
ISO 18193 Figure C.2 Vascular Model Overview

Definition and Purpose
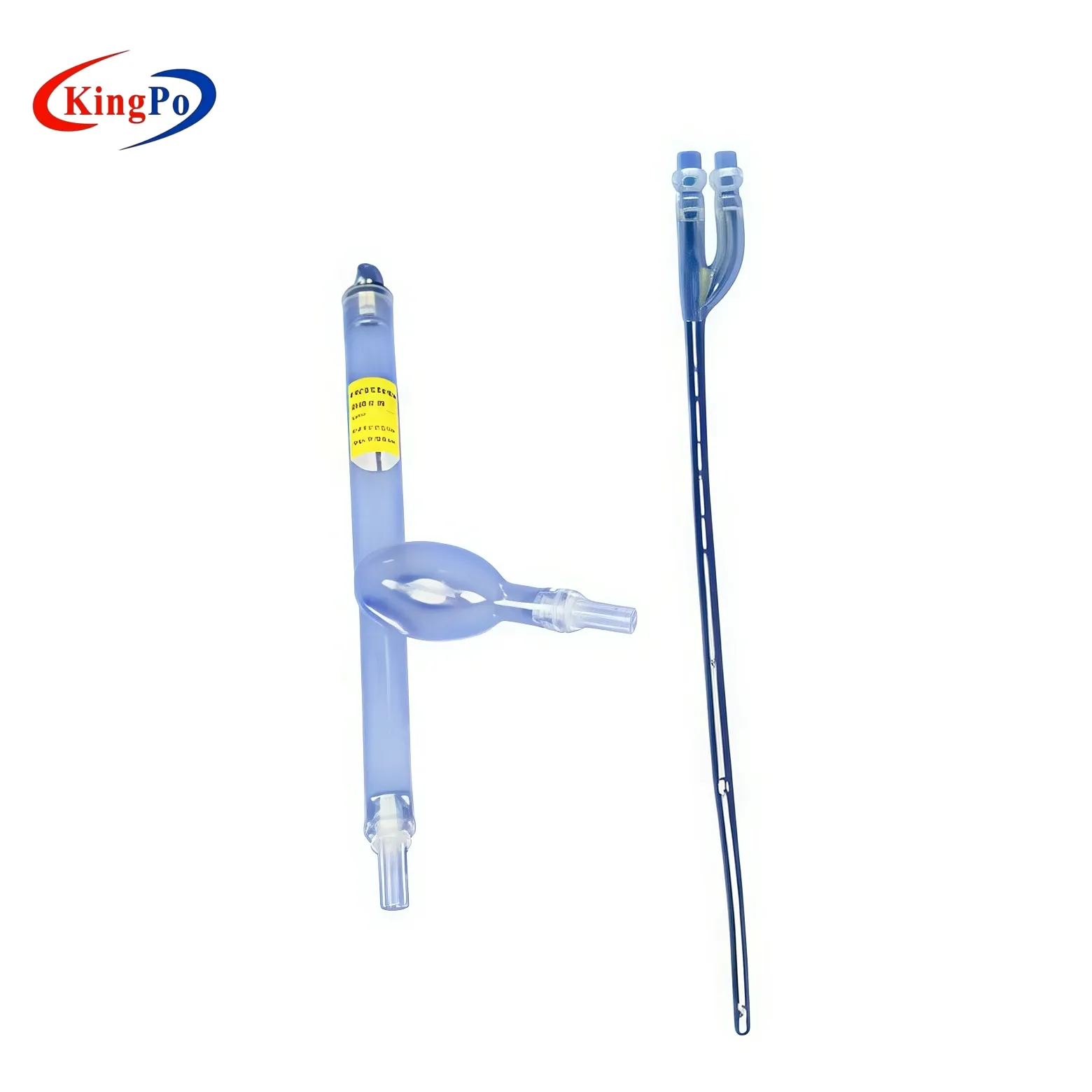
You use the iso 18193 figure c.2 vascular model to simulate the human cardiovascular system with high accuracy. This model helps you test dual-lumen cannulae in a controlled environment. The iso 18193 figure c.2 standard guides the design, ensuring that the vascular model matches the anatomy of the superior vena cava, right atrium, and inferior vena cava. You rely on this model to evaluate recirculation efficiency, which is essential for extracorporeal circulation systems. The iso 18193 figure c.2 vascular model supports your efforts in diagnosis, treatment, and prognosis of cardiac conditions. You can trust the model to deliver repeatable results for cardiac surgery, echocardiography, and the management of cardiac tumors. KingPo designed this model to meet strict iso 18193 figure c.2 compliance, making it a reliable tool for your laboratory or manufacturing process.
Discover the Vascular Model
Explore how the ISO 18193 Figure C.2 Vascular Model can enhance your testing processes and ensure compliance.
Key Anatomical Features
The iso 18193 figure c.2 vascular model includes precise representations of the superior vena cava, right atrium, and inferior vena cava. You see the anatomical fidelity in every curve and junction. The model’s transparent construction lets you observe flow patterns during testing. You can use ink or particles to visualize recirculation, which is vital for assessing dual-lumen cannula performance. The vascular model’s dimensions follow the iso 18193 figure c.2 requirements, so you get accurate simulation for cardiac diagnosis and treatment. You benefit from the model’s ability to mimic real vascular conditions, which supports your work in cardiac surgery, echocardiography, and the study of cardiac tumors. The model also aids in evaluating malignant cardiac tumors, helping you improve treatment strategies and prognosis.
Comparison with Other Models
When you compare the iso 18193 figure c.2 vascular model to other vascular models, you notice several advantages. Many models lack the anatomical detail required for iso 18193 figure c.2 compliance. The KingPo model stands out because it matches the exact specifications for vessel diameter, curvature, and junction angles. You get a model that supports advanced diagnosis, treatment, and prognosis for cardiac tumors and malignant cardiac tumors. Other models may not offer the same level of transparency, making it harder to observe flow during testing. The iso 18193 figure c.2 vascular model gives you clear visualization, which is crucial for cardiac surgery and echocardiography. You can trust this model to deliver consistent results, supporting your efforts in research, development, and quality control. The model’s design ensures that you meet iso standards for vascular testing, giving you confidence in your diagnosis and treatment planning.
Tip: Choose the iso 18193 figure c.2 vascular model for your cardiac device testing. You gain anatomical accuracy, compliance, and reliable data for diagnosis, treatment, and prognosis.
Technical Specifications and ISO Compliance
Materials and Construction
You rely on the iso 18193 figure c.2 vascular model for its advanced materials and construction. The model uses medical-grade transparent material that lets you observe flow patterns with clarity. You see every detail during testing, which helps you achieve certification for your devices. The model’s transparency supports visualization of ink and particles, making your recirculation tests more accurate. You benefit from a design that follows the iso standard, ensuring anatomical fidelity. The model replicates the superior vena cava, right atrium, and inferior vena cava with precision. You trust the construction because it meets strict manufacturing tolerances. The model’s durability allows repeated use in laboratory settings. You gain confidence knowing the model supports your certification and compliance needs.
Note: The transparent material in the model enhances your ability to monitor flow dynamics and verify device performance.
Precision and Calibration
You depend on the iso vascular model for its precision. The model features tight manufacturing tolerances, so you get consistent results every time. You use the model to calibrate your testing equipment and validate dual-lumen cannula performance. The calibration process follows iso guidelines, which helps you maintain compliance with certification requirements. You receive a model that undergoes rigorous factory calibration before delivery. You can trust the measurements because the model matches the iso standard exactly. You avoid errors in your testing process because the model provides reliable anatomical dimensions. You use the model to support product development and quality control. The model’s precision ensures you meet certification standards for medical devices.
|
Feature |
Benefit to You |
|---|---|
|
Tight tolerances |
Consistent test results |
|
Factory calibration |
Reliable measurements |
|
Anatomical accuracy |
Validates device performance |
ISO 17025 Traceability
You achieve lab accreditation and regulatory approval by using a model with iso 17025 traceability. The model’s traceability supports your compliance efforts and certification goals. You document every test with confidence, knowing the model meets international standards. You show regulators that your lab follows iso requirements for testing and calibration. The model’s traceability helps you maintain certification for your facility. You use the model to demonstrate compliance during audits and inspections. You rely on the model to support your application for market approval. The iso 17025 traceability gives you a clear path to certification and regulatory success.
Tip: Always choose a model with iso 17025 traceability to streamline your certification process and ensure compliance with global standards.
Testing Applications and Performance
Recirculation Testing for Dual-Lumen Cannulae
You use the iso 18193 figure c.2 vascular model to perform recirculation testing for dual lumen cannula devices. This vascular model gives you a clear simulation of the superior vena cava, right atrium, and inferior vena cava. You place the cannula in the vascular model and observe recirculation patterns. You rely on recirculation testing to measure how much blood returns to the cannula after passing through the circuit. You see the flow paths and identify areas where recirculation may affect device performance. You use ink or particles to visualize recirculation during testing. You repeat recirculation testing to ensure consistent results. You trust the vascular model to deliver accurate recirculation data for cardiac surgery and echocardiography. You use recirculation testing to improve diagnosis, treatment, and prognosis for patients with cardiac tumors. You rely on the vascular model to support recirculation testing for malignant cardiac tumors. You use recirculation testing to validate dual lumen cannula performance and optimize device design.
Tip: You achieve reliable recirculation testing by following the iso standard and using a vascular model with anatomical accuracy.
Compatibility with Extracorporeal Circuits
You connect the vascular model to extracorporeal circuits for recirculation testing. You use the iso 18193 figure c.2 vascular model because it fits standard extracorporeal systems. You test dual lumen cannula devices in a controlled environment. You observe recirculation and flow dynamics in the vascular model. You use the vascular model to simulate real cardiac conditions. You rely on recirculation testing to evaluate device performance in extracorporeal applications. You use the vascular model to support diagnosis, treatment, and prognosis for cardiac tumors. You trust the vascular model to deliver repeatable recirculation testing results. You use the vascular model for quality control and product development. You rely on the vascular model to meet iso requirements for extracorporeal testing. You use recirculation testing to improve cardiac surgery and echocardiography outcomes.
|
Application |
Vascular Model Benefit |
|---|---|
|
Extracorporeal circuits |
Accurate recirculation testing |
|
Cardiac surgery |
Reliable diagnosis |
|
Echocardiography |
Enhanced treatment planning |
|
Cardiac tumors |
Improved prognosis |
Use in CPB, ECLS, and ECCO₂R
You use the iso 18193 figure c.2 vascular model for recirculation testing in cardiopulmonary bypass (CPB), extracorporeal life support (ECLS), and extracorporeal carbon dioxide removal (ECCO₂R). You rely on the vascular model to simulate critical anatomical structures. You test dual lumen cannula devices under physiological conditions. You observe recirculation and validate device performance in clinical applications. You use recirculation testing to assess how the cannula works in CPB, ECLS, and ECCO₂R procedures. You trust the vascular model to deliver accurate recirculation data for cardiac surgery and echocardiography. You use recirculation testing to support diagnosis, treatment, and prognosis for cardiac tumors. You rely on the vascular model to evaluate recirculation in malignant cardiac tumors. You use recirculation testing to optimize dual lumen cannula design and improve patient outcomes. You follow the iso standard to ensure your testing meets clinical requirements.
Note: You achieve precise recirculation testing in CPB, ECLS, and ECCO₂R by using a vascular model that replicates the superior vena cava, right atrium, and inferior vena cava.
Quality Control and Product Development
You depend on the ISO 18193 Figure C.2 vascular model to strengthen your quality control process. This model gives you a reliable foundation for testing dual lumen cannula devices. You use the model to check recirculation patterns and verify cannula performance in extracorporeal circuits. You see how the model helps you detect inconsistencies during testing. You can identify issues early and prevent faulty devices from reaching patients.
You improve product development by using the model throughout the design cycle. You test prototypes and refine cannula geometry based on recirculation data. You observe flow dynamics and make adjustments to enhance performance. You use the model to simulate real-world conditions for extracorporeal applications. You gain valuable insights that guide your engineering decisions.
You follow a systematic approach to quality control:
-
Set up the model for testing dual lumen cannula devices.
-
Run recirculation tests in extracorporeal circuits.
-
Record performance data and compare results.
-
Analyze flow patterns and identify areas for improvement.
-
Adjust cannula design and repeat testing.
You rely on the model to support regulatory compliance. You document every testing step and show that your devices meet ISO standards. You use the model to generate objective data for market approval. You build confidence in your products by demonstrating consistent performance.
Tip: Use the model as a standard reference for all recirculation testing. You ensure that every dual lumen cannula meets strict quality requirements.
You collaborate with your R&D team to optimize cannula design. You share testing results and discuss ways to improve extracorporeal device performance. You use the model to validate new ideas and accelerate product development.
|
Step |
Benefit to You |
|---|---|
|
Testing with model |
Reliable recirculation results |
|
Data analysis |
Improved cannula performance |
|
Design refinement |
Enhanced extracorporeal safety |
|
Documentation |
Easier regulatory approval |
You trust the ISO 18193 Figure C.2 vascular model to deliver repeatable testing outcomes. You use it to maintain high standards in quality control and product development. You achieve better results and bring safer dual lumen cannula devices to the market.
Imaging and Visualization in Testing
Transparent Design for Flow Observation
You benefit from the transparent construction of the ISO 18193 Figure C.2 vascular model. This design lets you observe flow patterns directly during imaging. You can see how blood or test fluids move through the superior vena cava, right atrium, and inferior vena cava. The clear material supports both standard and advanced imaging techniques. You use this feature to monitor device placement and flow dynamics in real time. The transparent model also helps you compare results from different imaging methods. You gain a better understanding of how dual-lumen cannulae perform under various conditions. This clarity improves your ability to detect issues early in the testing process.
Note: Transparent models make imaging easier and more accurate, especially when you need to visualize complex flow paths.
Use of Ink and Particles
You enhance your imaging results by introducing ink or particles into the vascular model. These substances help you track flow direction and recirculation during testing. You can use different colors or particle sizes to simulate various clinical scenarios. This approach supports both single-mode and multimodal imaging. You combine visual observation with other imaging tools to get a complete picture of flow behavior. For example, you might use ink for direct visualization and pair it with multimodal imaging for deeper analysis. This method allows you to validate your findings across multiple imaging platforms. You improve your testing accuracy by using these visual aids.
-
You inject ink to highlight flow separation.
-
You add particles to observe turbulence.
-
You use multimodal imaging to compare results from different techniques.
Enhancing Data Accuracy
You rely on imaging to collect precise data during device testing. The vascular model’s design supports high-resolution imaging and multimodal imaging. You capture detailed images that show how the cannula interacts with the vascular structures. This information helps you measure recirculation rates and identify potential problems. You use multimodal imaging to cross-check your results and ensure consistency. By combining visual and quantitative data, you improve the reliability of your testing process. You also document your imaging results for regulatory submissions and quality control. Accurate imaging leads to better product development and safer medical devices.
|
Imaging Method |
Benefit to You |
|---|---|
|
Direct visualization |
Immediate feedback |
|
Multimodal imaging |
Comprehensive data analysis |
|
High-resolution scans |
Detailed flow assessment |
Tip: Always use multimodal imaging when you need to confirm your results and enhance your data accuracy.
Error Mitigation and Best Practices
Common Testing Pitfalls
You may encounter several challenges during testing with the ISO 18193 Figure C.2 vascular model. One common pitfall involves improper setup of the extracorporeal circuit. If you do not secure connections, leaks can occur and affect your results. Another issue arises when you use incorrect cannula placement. This mistake can lead to inaccurate recirculation data. You might also overlook calibration of your equipment, which can introduce measurement errors. Sometimes, you may rush the process and skip documentation. This makes it hard to trace results or repeat the procedure. You should also avoid using expired or contaminated ink and particles, as these can distort flow visualization.
Tip: Always check your setup and materials before starting any testing session.
Strategies for Reliable Results
You can achieve reliable results by following a few best practices. First, review the ISO 18193 standard before each testing session. This helps you stay aligned with the required procedures. Next, use a checklist to confirm that all connections are tight and secure. You should calibrate your equipment before every round of testing. This step ensures that your measurements remain accurate. Place the dual-lumen cannula carefully in the model, following the anatomical landmarks. You should also use fresh ink or particles for each test to maintain clear visualization. Record every step in a lab notebook or digital log. This habit helps you track your process and identify any deviations.
Here is a simple checklist for your reference:
-
Review the ISO 18193 standard.
-
Inspect and secure all connections.
-
Calibrate equipment.
-
Place cannula accurately.
-
Use fresh ink or particles.
-
Document every step.
Note: Consistency in your approach leads to more reliable testing outcomes.
Quality Assurance Measures
You can strengthen your quality assurance by implementing systematic controls. Start by training all team members on the correct use of the vascular model. Regular training reduces the risk of user error. You should schedule routine maintenance for your equipment and the model itself. This keeps everything in optimal condition. Use control samples during testing to verify that your system works as expected. You can also conduct peer reviews of your testing process. This allows you to catch mistakes early and improve your methods. Store your data securely and back it up regularly. This practice protects your results and supports regulatory compliance.
|
Quality Measure |
Benefit to You |
|---|---|
|
Team training |
Fewer user errors |
|
Routine maintenance |
Reliable equipment |
|
Control samples |
Validated results |
|
Peer review |
Improved accuracy |
|
Data backup |
Secure records |
Remember: Strong quality assurance ensures that your testing meets the highest standards.
Business Value and Support
R&D and Market Approval
You gain a strong advantage in research and development when you use the iso 18193 Figure C.2 vascular model. This model helps you test devices for cardiac tumors with precision. You can simulate real cardiac conditions and observe how your devices perform during surgery. The model supports you in every stage of product development, from early prototypes to final designs. You use it to collect data for diagnosis and treatment of cardiac tumors. You also rely on it to demonstrate compliance with iso standards. This evidence is essential for market approval. Regulators want to see that your devices meet strict requirements for cardiac diagnosis and treatment. You can show them clear results from imaging and recirculation tests. The model helps you document every step, making your approval process smoother.
Tip: Use the model to validate your device for cardiac tumors before you submit for regulatory review. This step saves you time and resources.
Procurement and Warranty
You can easily add the iso 18193 Figure C.2 vascular model to your lab or production line. The procurement process is straightforward. You receive a model that is ready for immediate use in cardiac diagnosis, treatment, and surgery. Each model comes with a full one-year warranty. This warranty covers any manufacturing defects. You can focus on testing cardiac tumors and improving treatment without worrying about equipment reliability. The model’s durability supports repeated use in imaging and diagnosis. You also benefit from clear documentation that comes with each unit. This documentation helps you set up the model for cardiac surgery and ensures you follow iso guidelines.
Here is what you receive with your purchase:
-
Fully calibrated iso 18193 Figure C.2 vascular model
-
One-year warranty for peace of mind
-
User manual for setup and imaging
-
Support for cardiac diagnosis, treatment, and surgery
Technical Support and Training
You receive ongoing technical support when you use the iso 18193 Figure C.2 vascular model. Expert teams help you with setup, imaging, and troubleshooting. You can contact support for questions about cardiac tumors, diagnosis, or treatment. Training sessions are available for your staff. These sessions cover best practices for imaging, diagnosis, and surgery. You learn how to use the model for cardiac tumors and improve your workflow. You also receive updates on iso standards and new imaging techniques. This support ensures you stay current in the field of cardiac diagnosis and treatment. You build confidence in your team’s ability to handle complex cases involving cardiac tumors.
|
Support Service |
Benefit to You |
|---|---|
|
Technical assistance |
Quick answers for imaging and setup |
|
Training sessions |
Improved skills in diagnosis and surgery |
|
Documentation updates |
Stay aligned with iso requirements |
|
Warranty coverage |
Reliable testing for cardiac tumors |
Note: Ongoing support and training help you achieve better outcomes in cardiac diagnosis, treatment, and surgery.
You gain clear advantages when you use the iso 18193 figure c.2 vascular model for vascular device testing. This model helps you achieve compliance and supports innovation in vascular product development. You see reliable results and improve your evaluation process. You meet high standards for vascular testing and build confidence in your lab. For more information, you can explore these resources:
-
Access the document here
FAQ
What is the main purpose of the ISO 18193 Figure C.2 vascular model?
You use this model to simulate human vascular anatomy for medical device testing. It helps you evaluate dual-lumen cannulae in standard extracorporeal test circuits and ensures your results meet ISO requirements.
How does the model support recirculation performance testing?
You rely on the model to visualize and measure recirculation. It lets you observe flow patterns and assess how much fluid returns to the cannula, which is essential for recirculation performance testing.
Can you use the model with all standard extracorporeal test circuits?
Yes, you can connect the model to standard extracorporeal test circuits. This compatibility allows you to test devices in realistic conditions and compare results across different setups.
What materials make up the vascular model?
You receive a model made from medical-grade transparent material. This construction lets you see flow dynamics clearly and supports repeated use in laboratory environments.
Why is anatomical accuracy important in device testing?
You need anatomical accuracy to ensure your test results reflect real human conditions. Accurate vessel shapes and sizes help you validate device safety and performance before clinical use.
How does the model help with regulatory approval?
You use the model to generate objective data for your submissions. It supports your compliance with ISO standards and helps you demonstrate that your devices work safely in extracorporeal circulation systems.
What is the basic testing principle when using this model?
You place the device in the model, run fluid through standard extracorporeal test circuits, and observe flow behavior. This testing principle helps you evaluate device function and recirculation.
Do you receive technical support with your purchase?
Yes, you get access to technical support and training. This service helps you set up the model, troubleshoot issues, and follow best practices for accurate testing.
Tip: Always review the user manual and reach out to support if you have questions about setup or testing.
ISO 18193 Figure C.2 Vascular Model | Dual Lumen Cannula Recirculation Testing







